Introduction
Label free MUC1 aptasensors based on electrodeposition of gold nanoparticles on screen printed electrodes. Anca Florea 1 , Zahra Taleat 1,2 , Cecilia Cristea 1 , Robert Săndulescu 1

Introduction
E N D
Presentation Transcript
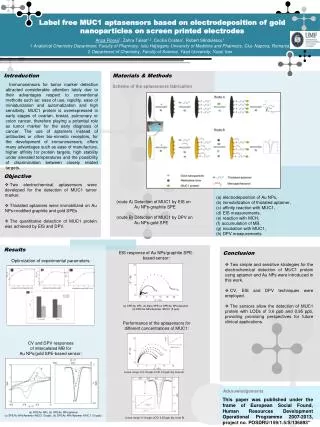
Label free MUC1 aptasensors based on electrodeposition of gold nanoparticles on screen printed electrodes • Anca Florea1, Zahra Taleat1,2, Cecilia Cristea1, Robert Săndulescu1 • 1 Analytical Chemistry Department, Faculty of Pharmacy, Iuliu Haţieganu University of Medicine and Pharmacy, Cluj- Napoca, Romania, • 2 Department of Chemistry, Faculty of Science, Yazd University, Yazd, Iran Introduction Materials & Methods Immunosensors for tumor marker detection attracted considerable attention lately due to their advantages respect to conventional methods such as: ease of use, rapidity, ease of miniaturization and automatization and high sensitivity. MUC1 protein is overexpressed in early stages of ovarian, breast, pulmonary or colon cancer, therefore playing a potential role as tumor marker for the early diagnosis of cancer. The use of aptamers instead of antibodies or other bio-mimetic receptors, for the development of immunosensors, offers many advantages such as ease of manufacture, higher affinity for protein targets, high stability under elevated temperatures and the possibility of discrimination between closely related targets. Scheme of the aptasensors fabrication Objective • Two electrochemical aptasensors were developed for the detection of MUC1 tumor marker. • Thiolatedaptamers were immobilized on Au NPs-modified graphite and gold SPEs • The quantitative detection of MUC1 protein was achieved by EIS and DPV. (a) electrodeposition of Au NPs, (b) immobilization of thiolatedaptamer, (c) affinity reaction with MUC1, (d) EIS measurements, (e) reaction with MCH, (f) accumulation of MB, (g) incubation with MUC1, (h) DPV measurements (route A) Detection of MUC1 by EIS on Au NPs-graphite SPE (route B) Detection of MUC1 by DPV on Au NPs-gold SPE Results Conclusion EIS response of Au NPs/graphite SPE-based sensor: Optimization of experimental parameters: • Two simple and sensitive strategies for the electrochemical detection of MUC1 protein using aptamer and Au NPs were introduced in this work. • CV, EIS and DPV techniques were employed. • The sensors allow the detection of MUC1 protein with LODs of 3.6 ppb and 0.95 ppb, providing promising perspectives for future clinical applications. . (a) SPE/Au NPs, (b) Bare SPE (c) SPE/Au NPs/Aptamer (d) SPE/Au NPs/Aptamer /MUC1 (5 ppb) Performance of the aptasensors for different concentrations of MUC1: CV and DPV responses of intercalated MB for Au NPs/gold SPE-based sensor: Linear range: 2.5-15 ppb; LOD: 3.6 ppb (by route A) Acknowledgements This paper was published under the frame of European Social Found, Human Resources Development Operational Programme 2007-2013, project no. POSDRU/159/1.5/S/136893” (a) SPE/Au NPs, (b) SPE/Au NPs/aptamer (c) SPE/Au NPs/Aptamer /MUC1 (5 ppb), (d) SPE/Au NPs/Aptamer /MUC1 (10 ppb); Linear range: 0-10 ppb; LOD: 0.95 ppb (by route B)