Catalyst
360 likes | 727 Views
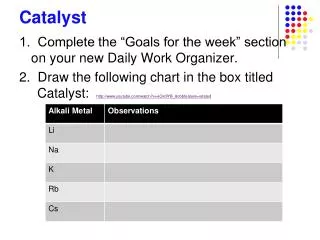
Catalyst. Put this Catalyst on the same sheet of paper you used yesterday. Check your Week 15 notes if you don’t know the answers! 1. Ionic bonds are between a _________ and a _________. 2. In covalent bonds, electrons are __________.

Catalyst
E N D
Presentation Transcript
Catalyst Put this Catalyst on the same sheet of paper you used yesterday. Check your Week 15 notes if you don’t know the answers! • 1. Ionic bonds are between a _________ and a _________. • 2. In covalent bonds, electrons are __________. • 3. Write the chemical formula for the ionic compound formed by K and N. • 4. What would be the name of this compound from #3?
Catalyst Get out a sheet of notebook paper and do the Catalyst! 1. All ionic compounds are named the same way, with two exceptions. What are these exceptions? (check your notes!) 2. Write the formula for the ionic compound formed by Li and S. (find stable charges and krisskross) 3. What is the name of the compound formed by Fe+2 and Cl-1? 4. What is the name AND chemical formula of the compound formed by calcium and sulfate. (you will need to check your notes for sulfate)
Exit Slip • 1. What is the correct formula and compound name for the compound formed by Ga and Cl? • GaCl, gallium chloride • GaCl3, gallium chlorine • GaCl3, gallium chloride 2. True or false: K and S would form potassium sulfide
Exit Slip 3. What is the correct name for the compound formed by Fe(II) and Br? 4. Write the correct name AND formula for the compound formed by Ca and At 5. What is the correct name for this compound: Al(OH)3? • Aluminum hydroxide • Aluminum oxide • Aluminum (III) hydroxide d. Aluminum hydroxide(III)
Naming Ionic Compounds BeCl2
Naming Ionic Compounds BeCl2 Barium Chloride
Naming Ionic Compounds Ag2S3
Naming Ionic Compounds Ag2S3 Silver (III) Sulfide
Naming Ionic Compounds Al2S3
Naming Ionic Compounds Al2S3 Aluminum Sulfide
Naming Ionic Compounds Rb3P Rubidium Phosphide
Naming Ionic Compounds Fe2O2
Naming Ionic Compounds Fe2O2 Iron(II) oxide
Naming Ionic Compounds Al3N3
Naming Ionic Compounds Al2N3 Aluminum Nitride
Naming Ionic Compounds Ba(CO3)
Naming Ionic Compounds Ba(CO3) Barium Carbonate
Naming Ionic Compounds Al(PO3)
Naming Ionic Compounds Al(PO3) Aluminum Phosphite
Naming Ionic Compounds H2(SO4)
Naming Ionic Compounds H2(SO4) Hydrogen Sulfate
Equation Form • EXAMPLE: Sodium and Carbonate
Equation Form • EXAMPLE: Sodium and Carbonate • Na+ + CO3-2
Equation Form • EXAMPLE: Sodium and Carbonate • Na+ + CO3-2 Na2(CO3)
Equation Form • EXAMPLE: Iron(II) and Chlorine
Equation Form • EXAMPLE: Iron(II) and Chlorine • Fe+2+ Cl-1
Equation Form • EXAMPLE: Iron(II) and Chlorine • Fe+2+ Cl-1 FeCl2
Exit Slip • 1. Which of the following is NOT a polyatomic ion? • CO2 • CO3-2 • PO3-2 d. SO4-2 2. What is the chemical formula for barium carbonate? a. Ba2(CO3) b. Ba(CO3)-2 c. Ba(CO3)
Exit Slip 3. What is the name of the following compound: H2(SO4)? a. Hydride sulfate b. Hydrogen sulfate c. Hydrogen sulfite 4. True or false: Aluminum(III) sulfide would be the correct name for Al2S3 5. Write the chemical formula for iron(III) sulfide