Real Time RT PCR
Real Time RT PCR . Broad and Long Term Objective.

Real Time RT PCR
E N D
Presentation Transcript
Broad and Long Term Objective To characterize the expression of ribulose 1-5 bisphosphate carboxylase oxygenase and chlorophyll AB binding gene in Lycopersicon esculentum (Tomato) leaves subjected to either 48 or 72 hours in the dark as compared to the expression in leaves grown under normal 12 hr light/dark cycle and harvested at noon.
Research Plan RNA Isolation leaf material grown in light and in the dark RNA Electrophoresis and cDNA synthesis Assessing Gene Expression Northern Blot RNase Protection Quantitative PCR Quantitative real time RT PCR
Electrophoresis of RNA and cDNA Intact High Quality RNA Characterized by: • Two prominent rRNA Bands • Slight smear of various sized mRNA molecules in background • When isolating RNA from leaf material, may see plastid rRNA bands cDNA Synthesis Products Characterized by: • A smear of many different sizee transcripts between .5-3.0 Kb
Today’s Laboratory Objectives To understand the theoretical basis behind primer design for real time RT-PCR analysis To become familiar with the manner in which a real time RT PCR experiment is set up and the data is collected
Primer Design Primer Design Parameters • Targets an amplicon length of 75 to 150 bp • 50 to 60% GC content • Limit secondary structure • Limit stretch of G or C’s longer than 3 bases • No stable interaction between forward and reverse primers • Place C’s and G’s on ends of primers (no more than 2 inlast 5 bases on 3’ end) • Melting Temperature (Tm) above 50 °C (70-72° C) • Verify specificity
Experimental Design Relative Expression • Samples: 1) cDNA derived from RNA from leaves at 48 or 72 hour in dark 2) cDNA derived from RNA from leaves harvested at 12 noon 3) no template control • Each experimental sample assayed in triplicate • No template controls assayed in duplicate • Total of 8 samples/group • Dilute cDNA 1/100 • Prepare a Master Mix for 9 Rxns Reaction Mix cDNA (diluted 1:100) 5.0 μl Sybr Green Super Mix 7.1 μl x 9 = 63.9 ul Primer A (20 μM) 0.3 μl x 9 = 2.7 ul Primer B (20 μM) 0.3 μl x 9 = 2.7 ul dH2O 12.3 μl x 9 = 110.7 ul total volume per reaction 25 μl x 9=
RubisCO Small Subunit accession #X05986 Coding Sequence 1 aaaaatgaaa aactcgtcag aaagaaaaag caaaagcaac aaaaaaattg caagtatttt 61 ttaaaaaaga aaaaaaaaac atatcttgtt tgtcagtatg ggaagtttga gataaggacg 121 agtgaggggt taaaattcag tggccattga ttttgtaatg ccaagaacca caaaatccaa 181 tggttaccat tcctgtaaga tgaggtttgc taactctttt tgtccgttag ataggaagcc 241 ttatcactat atatacaagg cgtcctaata acctcttagt aaccaattat ttcagcaatg 301 gcttcttcag taatgtcctc agcagctgtt gccacccgcg gcaatggtgc acaagctagc 361 atggttgcac ccttcactgg actcaagtcc accgcttctt tccctgtttc aaggaagcaa 421 aaccttgaca ttacctccat tgctagcaac ggtggaagag tcagttgcat gcaggtttgt 481 gtgtgtatat atatatacgt acaacaaaat tcattgacta taatgttata ctcgattagc 541 taatttaact atttataatt gtataggtgt ggccaccaat taacatgaag aagtacgaga 601 ctctgtcgtaccttcctgatttgtccgacg agcaattgct cagcgaaatt gagtacctat 661 tgaaaaatggatgggttccttgcttggaat tcgagactga ggtcaacatc tatctcctct 721 gtttttaaaa tttactagct agtatgttga tatgtcgtgt taacagtgtt gtgggatatc 781 atgtgcagca cggatttgtg taccgtgaga accataagtc accaggatac tacgatggca 841 gatactggaccatgtggaag ttgcccatgt tcgggtgcactgatgcaacccaggtcttgg 901 ctgaggtgca ggaggcaaag aaggcttacc cacaggcatg ggtccgtatc atcggattcg 961 acaatgttcg tcaagtgcag tgcatcagtt tcatcgctta caagcccgaa ggatactaaa 1021 tgtgtatatg tcaacagtga gaaactgttc gcattttccg ttttgcttct ttctttctat 1081 tcaatgtatg ttgttggatt ccagttgaat ttattatgag aactaataat aatagtaata 1141 atcatttgtt tctttactaa tttgcatttt cacatatgat ttctggtgca tatcataatt 1201 ttcattccac caatattaat ttccccattc aagttactta tgaaatagaa atcctcttct 1261 ccgactactt tatttgtccg aaagtcttgt ggctgctata taacgcaaaa tggatagaga 1321 agattcatta ctaagccgat c
RubisCO Primer Sets StartLengthTm%G+CSeq Primer Set A Left Primer 276 20 60.69 45 AAATGGATGGGTTCCTTGCT Right Primer 422 20 59.58 50 AAGACCTGGGTTGCATCAGT Product Size: 147 Primer Set B: Left Primer 216 22 59.87 50 GTCGTACCTTCCTGATTTGTCC Right Primer 375 20 59.96 55 GGTCCAGTATCTGCCATCGT Product Size: 160
Chlorophyll A/B Binding Protein (CAB-1b) Accession # M14443 Coding Sequence 1 atgaagaagt tgatggatta tagattgcca agtgtgctac acatgggatc ttgataccca 61 atgagatcat acatatagat atcacttgat aagatgattc tctctctttt ctcctatata 121 ttctcaaccc caactaactt catcttcatc acccatcaaa cacttaattc ttctcttaaa 181 ataaacacaa atggcagctg ctacaatggc tctttcttcc ccttcatttg ctggacaggc 241 agtcaaactctcaccatctgcctcagaaat ttctggaaat ggaaggatca ctatgagaaa 301 ggctgttgcc aagtccgccc catctagcag cccatggtat ggccctgacc gtgttaagta 361 cttgggccca ttctctggtg agtccccaag ctacttgacc ggtgaatttcctggtgatta 421 cgggtgggat accgctggac tttcagcaga ccctgaaact tttgccaaga accgtgaact 481 tgaagtgatc cactgcagat gggctatgct tggtgctctt ggatgtgtct tccctgagct 541 cttggcccgt aatggtgtca agttcggtga ggctgtgtgg ttcaaggccg gatcccagat 601 cttcagtgaa ggtggacttg actacttggg caacccaagc ttggtccatg cacaaagcat 661 cttggccatc tgggcttgcc aagttgtgtt gatgggagct gttgagggtt accgtattgc 721 tggtggacct cttggtgagg ttgtcgaccc actctaccct ggtggcagct tcgacccatt 781 aggccttgct gaagacccag aggcatttgc tgagctcaag gtaaaggaga tcaagaacgg 841 tagacttgct atgttctcta tgtttggatt ctttgttcaa gctattgtca ccggaaaggg 901 tccattggag aaccttgctg atcaccttgc agaccccgta aacaacaatg cctgggcttt 961 cgccacaaac tttgtccccg gaaaatgact ctaaacgtct caagtcttgg tcgtttgatg 1021 acagtgtaaa gatgtagtgt gctacctgac aatataatga aattttgttt gtgtttgaat 1081 ggcttttctg tactgagttt cattttccca agtcaactca taaatcaagc actaacaatg 1141 atacaacaaa atgacccctc acatatgagt aataactaga aaaactgcaa tgctatgttg 1201 taaggttgaa cttgaatttt caactagagc agtttattta atttaatgaa ttc
Chlorophyll A/B Binding Protein Primer Set StartLengthTm%G+CSeq Primer Set A Left Primer 55 21 59.86 47.62 AAACTCTCAACCATCTGCCTCA Right Primer 236 20 60.74 50 CACCCGTAATCACCAGGA Product Size: 147
Real Time RT PCR Real Time PCR Work Flow Sample---RNA Isolation---cDNA Synthesis---RT PCR Amplification
Real Time RT PCR Cycling Parameters Polymerase activation 95° C 10 min 40 cycles Denaturation 95° C 60 sec Primer Annealing 60° C 30 sec Extension 72˚ C 45 sec Melt Curves Denaturation 95° C 1 min Renaturation 55° C 1 min Denaturation Ramp 0.5° C every 10 sec
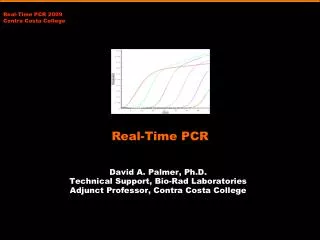
Defining Parameters ofReal Time RT PCR Cycle Threshold: Cycle # when product fluoresence exceeds that of background Fold Change: 2ΔCt Melt Curve: fluorescence plotted as a function of temperature as thermal cycler heats through dissociate temperature of product
Presentation of Real Time RT PCR Data What to include: • Melt Curve • Raw Quantitative Graph • Histogram of Relative Fold Change
Next Lab Dec 5th Real Time RT PCR Data Analysis using ICycler Software