TEM
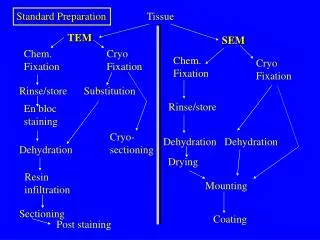
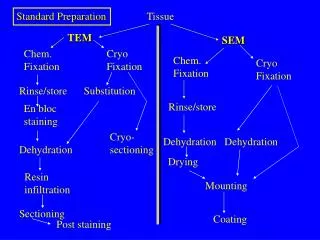
Standard Preparation. Tissue. TEM. SEM. Chem. Fixation. Cryo Fixation. Chem. Fixation. Cryo Fixation. Rinse/store. Substitution. Rinse/store. En bloc staining. Cryo- sectioning. Dehydration. Dehydration. Dehydration. Drying. Resin infiltration. Mounting. Sectioning.

TEM
E N D
Presentation Transcript
Standard Preparation Tissue TEM SEM Chem. Fixation Cryo Fixation Chem. Fixation Cryo Fixation Rinse/store Substitution Rinse/store En bloc staining Cryo- sectioning Dehydration Dehydration Dehydration Drying Resin infiltration Mounting Sectioning Coating Post staining
Dehydration • Reasons for dehydration: • Water in incompatible with conditions inside an electron column. • Most of the materials used to infiltrate and embed specimens prior to ultrathin sectioning are hydrophobic. • Methods of Dehydration: • Organic solvent Series • Tissue is transferred through a series of organic solvents in increasing concentration. • Ethanol and acetone are the most commonly used. • Water content is slowly reduced to the point that the tissue is in 100% solvent. and is thus completely dehydrated.
Embedding and Sectioning • Requirements for cutting any material into thin slices: • Support - biologicals tend to be soft. Inducing hardness in them gives them the mechanical support needed for sectioning. • Accomplished by lowering temperature (freezing) or infiltration with some material that can be hardened. • Plasticity - resiliency as opposed to brittleness.
Embedding and Sectioning • Cryosectioning • Commonly done for light microscopy. • ie hospital operating room biopsies. • Rapid. • Preservation is usually sufficient for a rapid diagnosis. • Overall resolution is low. • Ultrathin cryosectioning • Technically demanding • Requires expensive specialized equipment • Ultrastructural preservation often poor due to freezing artifact. • Usually done only when tissue cannot be exposed to chemical fixatives...as in some immunolabeling, analytical work.
Embedding and Sectioning • Embedment • Light microscopy • Tissue infiltrated with molten paraffin wax - which is allowed to cool and harden. • Requires dehydration and infiltration with a paraffin solvent - aromatic hydrocarbon (xylene, toluene, benzene). • Provides sufficient support to section to about 3 micrometers minimum with a steel knife. • Paraffin can infiltrate deeply into tissue, allowing large blocks and ultimately large sections to be obtained.
Embedding and Sectioning Paraffin Sectioning for Light Microscopy
Embedding and Sectioning • TEM Embedment • Tissue infiltrated with a resin which is polymerized by heat, chemicals, or U.V. • Provides support to section infiltrated tissue to about 40 nm minimum. • Infiltration is limited...specimens can be no more than a few mm thick. • The required thinness of the sample and the friction during cutting limits the section size to about 1 mm2 maximum.
Embedding and Sectioning • Types of Resins • Acrylics - ie methyl, butyl methacrylates (plexiglass) - "Open-structured" - allows for better stain penetration and Antibody rxn • Epoxies - epon, araldite, Quetol, Spurr - for most general work • Polycarbonates - vestopal - fiberglass resin • Epoxy Resins - most commonly used. • Components: • Resin - Epon 812, Araldite 502 or 6005 • Hardener - DDSA - amount can be varied • Plasticizer - NSA • Accelerator - DMP-30
Embedding and Sectioning • Infiltration • In resin/solvent mixture in increasing concentration • Ethanol/resin or acetone resin often used • Propylene oxide/resin is most effective • When 100% resin is reached, it should be changed twice to insure that all solvent is removed • Polymerization • Thermal - 50-70 C, depending on resin mix • U.V. - usually done to avoid heat • of polymerization. Often done at low temp.
Embedding and Sectioning • Ultramicrotomy • Mechanical Advance • Thermal Advance • Ultramicrotome Knives: • Diamond - 1.5 - 6mm cutting edge • Latta-Hartmann (glass) - 6mm cutting edge (~1mm useable) • Both use water to support and lubricate the section as it is cut (decreases friction)
Embedding and Sectioning • Making a glass knife: • Use of a glass knifemaker to score a 1" glass square
Embedding and Sectioning A scored 1" glass square (top) and the resultant glass knife: Making the water trough Tape or plastic a) Cutting edge b) Knife angle (45o) c) Corner d) Shelf
Evaluating a glass knife - factors to consider: • Age - degrade rapidly due to edge flaking • Quality of cutting edge - flat, concave, convex • Amount of cutting edge - judged by the stress line. A "spur" is normal. • Contamination - on edge or sides.
Setting up the Microtome Block face Sample Block Knife edge Glass Knife
Tools Needed: Syringe - adjusting water in trough Loop - assist picking up sections Eyelash tools - assist with section manipulations
Sectioning - Troubleshooting • Factors affecting ultrathin sectioning quality: • Embedment - poor infiltration, polymerization, too soft, too hard, brittle, etc. • Quality of Knife - sharpness, scratches • Dullness - alternate cutting and skipping. • Compression - lines perpendicular to the direction of cut. • Microgrooves • not resolvable by LM • in their absence, the knife edge will appear as a bright line under a dissecting scope • cause striations parallel to the direction of cut
Sectioning • Factors affecting ultrathin sectioning quality: • Contamination (of knife, specimen block, or trough water) - oil, dust. Can cause lines or seen on sections • Knife angle • Usually 4o - 6o • too low=compression; too high=chatter • Environmental factors: • Building vibration - antivibration measures (inner tubes, tennis balls, granite slabs, etc…) • Static electricity - usually causes sections to be pulled down the back of the knife. • Try grounding the microtome, increasing humidity in room, or use a Zerostat or Staticmaster
Sectioning More factors affecting sectioning quality: • Wind currents • Cutting speed - must be fast enough that vibration does not cause uneven cutting (chatter) and slow enough that the section is not compressed. • Water level in trough… • too high = block wetting; too low = compression • Block size and shape - the trapezoid.
Embedding and Sectioning • Section Thickness • Ideally, sections should be in the 55 - 60 nm range. • This allows for enough stain uptake for contrast, and maximum resolution (limited in the TEM by specimen-induced chromatic aberration). • Determined by interference colors. • Maximum thickness should not exceed 85 - 90 nm (light gold). • Thickness can sometimes be reduced by one color range by flattening sections - smooths out compression to a limited extent. Toluene, xylene, chloroform, heat.
Section Mounting • Specimen Grids • 3 mm support for TEM specimens. (a few are 2.6mm) • Different materials..usually copper...also nickel, gold, aluminum, platinum, stainless steel, beryllium, carbon, nylon. • Most are manufactured individually by electroplating; some are punched from screen stock; a few are woven. • Also differ by mesh size (bars per inch) 0 - 1000m • The smaller the mesh size, the greater the support (section drifting, splitting), but the less open area for viewing.
Section Mounting • A 200m grid has 60% open area; a 400m grid only 40% • Thin-bar grids...more fragile, more expensive. • Ultrathin sections can be supported on a bare grid of no greater than 200m. • Commonly used TEM grid types:
Picking up sections Mesh grids Eyelash tool Slot grids
Collecting on slot grids Dried on bridge, then punched out for viewing Sections floating on water
Section Mounting An ultrathin section on a 50m support filmed grid at 200X mag.
Post-Staining • Normally done, even if enbloc staining (ie uranyl acetate) has been done. • Uranyl acetate - 0.5 - 2% aqueous or saturated ethanolic or methanolic • Lead citrate - several formulations (Venable and Coggeshell; Reynolds) mostly using lead nitrate chelated with sodium citrate. • Adequate rinsing between and after staining is essential to prevent post-stain contamination. • Particular care must be used to exclude CO2 to inhibit lead carbonate formation - black cannonballs.
Contrast • Light Microscopy: • Contrast achieved by: • Use of special optics and filters which impart selective colors or brightness to areas differing in thickness or composition. E.g. - phase contrast, D.I.C. optics.
Contrast • Light Microscopy: • Contrast achieved by: • Selective staining • Chromatic stains selectively bind to specific components in the specimen. E.g. - Hematoxylin/Eosin
Contrast • Transmission Electron Microscopy: • Contrast is produced by the adsorption of heavy metals to specimen macromolecules. • The ability of an atom to absorb electrons is directly related to its mass. • Since biological specimens are composed mostly of low atomic # elements (C,O,H,N), they lack endogenous contrast....thus contrast is induced by "staining" with heavy metals. • Microscopists refer to the measure of a specimen's ability to absorb electrons as its electron density (vs electron “transparency”).
Contrast • Transmission Electron Microscopy: • Heavy metals commonly used for contrasting in TEM: uranium, lead, osmium, ruthenium, molybdenum, gold, silver. • It is the differential adsorption of various heavy metals to tissue components that produces the electron image of biological thin-sectioned materials. • The image may be composed of areas ranging from completely black to completely white with all ranges of grey in between. • Images with mostly pure blacks and whites are "contrasty" images, while those containing mainly greys are "flat” images.