Comenius University Bratislava Aquaculture Vaccinology William (Bill) W. Kay, PhD. Prof. ( Emeritus ), Dept. of Bioche
490 likes | 887 Views
Comenius University Bratislava Aquaculture Vaccinology William (Bill) W. Kay, PhD. Prof. ( Emeritus ), Dept. of Biochemistry & Microbiology University of Victoria, Victoria, B.C. CEO & Pres. Microtek International Inc., Saanichton, B.C. A life of swimming in a changing cocktail of

Comenius University Bratislava Aquaculture Vaccinology William (Bill) W. Kay, PhD. Prof. ( Emeritus ), Dept. of Bioche
E N D
Presentation Transcript
Comenius University Bratislava Aquaculture Vaccinology William (Bill) W. Kay, PhD. Prof. (Emeritus), Dept. of Biochemistry & Microbiology University of Victoria, Victoria, B.C. CEO & Pres. Microtek International Inc., Saanichton, B.C.
A life of swimming in a changing cocktail of microorganisms –some of which are highly virulent pathogens. Recombinant subunit vaccines
What are the commercially important finfish species? Salmonids: Atlantic salmon, Rainbow trout, Pacific salmon (Coho, Chinook). Non salmonids: Tilapia sp.,Channel Catfish, Striped Bass, Sea bass, Carp sp. (Koi, Grasscarp), Sablefish, Cod, Halibut, Turbot, Eel, Seabream, Seabass, Yellowtail sp., Ayu, Hamachi, Barrumundi, Milkfish, Japanese amberjack, Bluefin Tuna, Cobia etc…. It is estimated there are >300 cultivatable finfish sp. (…and they all get sick!)
What are the commercially important microbial pathogens of fish? Bacteria:Vibrio sp: (V.anguillarum Types I&II, V. salmonicida, Vibrio viscosus [Moritella viscosa]); Aeromonas sp: (A. salmonicida, A. hydrophila, A. cavia); Flavobacterium sp: (F. psychrophilum, F. columnare, F. maritimus [Tenacibacterium maritimum]); Edwardsiellasp: (E. ictaluri, E. tarda);Streptococcus sp. (S. iniae, S. agalactiae); Mycobacterium sp. (M. marinum, M. fortuitum, M. chelonae); Pseudomonas sp. (P. plecoglossida, P. anguiloseptica) Others:Yersinia ruckerii, Renibacterium salmoninarum, Piscirickettsia salmonis, Francisella sp., Photobacterium damselae, Lactococcus garvieae.………ad nauseum.
pathogens (cont.)Viruses: Family: • IHNV* (Infectious Hematapoietic Virus) Rhabdoviridae • VHS (Viral Hemorrhagic Septicemia) Rhabdoviridae • IPNV* (Infectious Pancreatic Necrosis Virus) Birnaviridae • ISAV* (Infectious Salmonid Anemia Virus) Orthomyxoviridae • PDV (Pancreatic Disease Virus) Togaviridae • CCV (Channel catfish virus) Herpeviridae • KHV (Koi Hepes Virus) Herpeviridae • CPV (Carp Pox Virus) Hepesviridae • IRV (Iridovirus) Iridoviridae • NDV* (Nodavirus Disease) Nodaviridae • HSMI (Heart & Skeletal Muscle Inflamation Virus ) ???
Pathogens (cont.)Parasites: Endoparasite: Disease Family • Kudoa thyrsites Soft Flesh Disease Myxosporean • Ceratomyxa shasta Ceratomyxosis Myxosporean • PKX Pancreatic Disease Myxosporean • Myxobolus cerebalus Whirling Disease Myxosporean Ectoparasite: • Gyrodactylus salaris GyrodactylosisMonogean • Lepeophtheirus salmonisSea lice Caligean • Caligus sp. (C. elongatus, C. teres, C. rogercressyi, etc.)
What is a Vaccine? • Anything (antigen) derived from a pathogen that when administered to a target animal (fish): • elicits a specific protective response against a foreign body = IR (immune response) • specific response must also have a memory component = DOI (duration of immunity)
How do we administer vaccines to fish? • IP or IM injection (by hand or machine) • Immersion. • Oral: • Dried with feed • encapsulated
Traditional vaccine testing & clinical trials: The “White Box” (i.e. The Wetlab) Direct immersion Oral Lethal Challenge giving rise to RPS or SL attachment The ‘Black Box’ (i.e.The “Field” ) Injection
LD50 or Relative Percent Survival (RPS) How do we measure efficacy? RPS = {1- (%Mort. Vaccinates/%Mort. Controls)}100 RPS 75 85 96 100
Aquaculture Vaccines - History • Furunculosis - Proof of concept - Duff (1942) • Commercial bacterial vaccines (mid 1970’s) • Oil adjuvanted injectable vaccines (early 1990’s) • Commercial viral vaccines (mid 1990’s) • Commercial recombinant vaccines (late 1990’s) • Commercial DNA vaccine (2005) • Reverse engineered & multiepitope vaccines (2007-)
Impact of vaccines on salmon culture From Markestad and Grave, 1997
Whole organism Traditional vaccine development or….“Grow ‘em and Show ‘em” Virus Formulations with Adjuvant(s), emulsions, stabilizers, preservatives etc. Chemical inactivation & processing Ex.Advantigen 5.1 Bacteria
Advantigen 5.1 EU Serial Release - 3 serials: CEFAS * Suggested figure - no monograph standard.
Complications of whole cell vaccines • Selection of representative strains • Maintenance of immunogenicity • Safety and potency • Interference • Inactivations and formulations may differ • QC/QA issues • Shelf life issues • Regulatory Issues • Cost of goods
Complications to multivalent vaccines • Reproducible disease models must be established for each disease ($$$$). • Intermittent fish availability is a problem. • Indirect disease criteria are difficult to convince regulators. • COGs is high since separate fermentations are required for each component. • EU GMP (human vaccine) production standards.
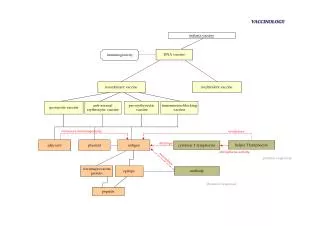
Envelope G protein Matrix proteins (M1 & M2) Core: L = polymerase N = nucleocapsid Fimbriae LPSs OMP Flagella Capsules or S-Layer How has biotechnology changed this?(Targeting specific antigens for “subunit” vaccines) Ex. viruses vs bacteria or ~5 vs ~50 targets. Secreted factors
Classical subunit vaccines Polysaccharides and proteins, purified from pathogenic organisms, and detoxified toxins, are examples of subunit immunogens. The subunits of pathogenic origin are safe to use as vaccines, provided that extraction procedure or detoxifying methods gives a pure product. Production of subunit vaccines generally requires large-scale, complicated down-stream processing of pathogenic organisms. These vaccines need adjuvants or various conjugates to render them more immunogenic.
Recombinant subunit vaccines – demystified. Cut DNA ExtractDNA Insert gene into a plasmid Pathogen Amplify in E. coli Produce in E. coli E.coli E. coli Introduce into E. coli Formulate Ex.Sea lice proteins made by harmless, boring, old E. coli
Upside of recombinant subunit vaccines • Pathogens can be entirely excluded from production = safety. • Multi-epitope vaccines engineered to a single strain = lower COGs. • Suppressor genes can be eliminated = detoxified. • Immune response can be tuned with pTCEs = more efficacious. • Better antigens can be further engineered for ex. through invitro evolution = more efficacious. • Vaccines have longer half-lives and can be stored frozen prior to formulations (clock starts at formulation) = lower COGs. • Genome sequencing projects provide advance information= more antigens.
Downside of recombinant subunit vaccines • The development is time consuming and more expensive than live attenuated or killed vaccines - ($$$$). • Requires highly skilled (expensive) R&D teams - ($$$). • Some regulatory agencies are slow to accept rDNA products - (time & $$$$). • Multiple antigens may be required - ($$$$).
Recent subunit vaccines • Via Recombinant DNA Technology: • Protein Engineering • Nucleic Acid Vaccines Antigenic fragment from A. Salmonicida OMP. OMP from E. coli
A Model Case:Piscirickettsia salmonis • A gram negative, rickettsia bacterial pathogen • Aetiological agent of salmonid rickettsial septicaemia (SRS) • First recognized and isolated in 1989 from a coho salmon (Oncorhynchus kisutch) • SRS afflicts global aquaculture of salmon • A devastating problem in Chile, emerging in NA & EU.
Salmonid Rickettsial Septicaemia (SRS) • P. salmonis causes a systemic infection • obligate intracellular pathogen • up to 40% of net pen stock can be lost annually ($150M-200M in Chile) • SRS is poorly managed by antibiotic treatment • natural reservoir or vector is unknown
Growth of P. salmonis • grown in salmonid cell culture @15ºC • 14-18 days for full growth • exceptionally low yields (~1/10 other Rickettsias) • grows within cytoplasmic vacuoles of host cells
Large scale growth ofP. salmonis • A very large monolayer surface area is required • 3 months minimum from freezer to harvest. • More complex than growing viruses.
Genomic DNA library construction • Isolated P. salmonis DNA free of host cell chromosomal DNA contamination. • Obtained ~250 µg DNA from 12,000 cm2 purification • Constructed expression library in λZAP II
Library screening: • Goal: to identify immunoreactive clones • Only available tool for screening: • rabbit Pabs • Salmon Pabs • Obtained several strongly immunoreactive clones
Trialing Putative SRS Vaccines Weak Challenge Poor RPS and DOI
Protein Engineering: Introducing Immunostimulatory Peptide Sequences Two different immunostimulatory sequences were added to the original OspA fusion protein resulting to three constructs: pET-CM17E2 pET-CT17E2 pET-CMT17E2
Immune response to promiscuous T-cell epitopes in OspA. Stimulation Index (SI) = ratio of specific stimulation (Pvacc/Pcon)
Potency of the 3rd generation Bayovac SRS RPS up to 100%
1 - preinduced sample 2 - 1 hour of induction 3 - 2 hours of induction 4 - 3 hours of induction The Bratislava connection -Large scale production of a recombinant SRS vaccine A B Jan Burian, Ph.D. VP R&D Microtek.
Other applications of this technology • Viruses: IHNV, IPNV, ISAV • Flavobacterial diseases • Kudoa thyrsites • Sea lice (Lepeotherius salmonis & Caligus rogercresseyi)
A Subunit vaccines for parasitic diseases of salmon Endoparasite B B Ectoparasite Kudoa Parasiticide control is costly and environmentally questionable. Concern over developing resistance. Concern over transmission to wild species. Vaccine control was previously considered to be impossible. Sea lice
Discovering sea lice antigens • Proteomics and genomics approaches to reveal target antigens. • Target genes subcloned and DNA sequenced. • Cloned target antigens expressed and formulated for trials SDS PAGE of SL Clones Immunoblot of SL Clones
Testing of prospective Sea Lice antigens • 3 Targets/19: • Attachment • Fertility • Feeding Control These 3 targets have been engineered into a trivalent single vaccine and is in trials.
Summary • Aquaculture vaccines are available for a variety of • microbial diseases. • Vaccines can be comprised of monvalent-multivalent • components which engage the immune system of fishes • Genetic engineering has enhanced our capacity to • design effective vaccines. • New-age vaccines for a variety of previously intractible • diseases are now becoming available. • Vaccines for sea lice and ISAV are now within reach • and close to commercial development.
The Current Microtek Team Research and Development William Kay, PhD Jan Burian, PhD Sharon Clouthier, PhD Eric Anderson, PhD Elizabeth Crump, PhD Joe Barlow, BSc; Kyle Clarke, BSc; Chuong Hyunh, BSc, Jan Burian Jr. BSc. Development , Production & Regulatory Steve Carlos, BSc Emily Coble, BSc ADA Sales and Marketing Patty Byrne, BSc, ADA Brandi Tudor, BSc. Fish Health Services Tim Hewison, BSc Hernan Pizarro, BSc Jesse Kelter, BSc. MSc. Wetlab: Norm Johnson, BSc., Stephanie Scheraga, BSc.
The Historical U. Victoria Team • Trevor Trust, Ph.D. • Bill Kay, Ph.D. • Ed Ishiguro, Ph.D. • Terry Pearson, Ph.D • Bob Olafson, Ph.D. • Fran Nano, Ph.D. • Innovation and Development Centre (Technology Transfer) Happiness is a healthy fish!
The Microtek Team Research and Development William Kay, PhD Julian Thornton, PhD Jan Burian, PhD Sharon Clouthier, PhD Eric Anderson, PhD Michael Kuzyk, PhD Elizabeth Crump, PhD Shannon Balfrey, PhD Dave Machander, MSc. John Drennan, MSc Daphne Dolhane, BSc. Kathy Gurgul, BSc. Iqbal Kathrada, BSc. Development and Production Steve Carlos, BSc Patty Byrne, BSc, ADA Tim Hewison, BSc Steven Gale Fish Health Services Joe Barlow, BSc Emily Coble, BSc Hernan Pizarro, BSc Steve Cameron, MSc. Wetlab: Melanie Sheppard, BSc, ADA Mike Norris, BSc, ADA Joe Klimik, BSc. MSc.