Boiling Points
Boiling Points. Andrew, Annie, Paulina, Claudia. What Is Boiling Point?. The temperature at which the vapor pressure of the liquid equals the environmental pressure surrounding the liquid.

Boiling Points
E N D
Presentation Transcript
Boiling Points Andrew, Annie, Paulina, Claudia
What Is Boiling Point? • The temperature at which the vapor pressure of the liquid equals the environmental pressure surrounding the liquid. • When a substance boils, most of the remaining attractive forces are broken so the particles can move freely and far apart. The stronger the attractive forces are, the more energy is needed to overcome them and the higher the boiling temperature. • Units: Celsius (⁰C) or Kelvin (K)
Elements at the center of the periodic table have the highest boiling/melting point • The non-metal carbon possesses the highest boiling point of all the elements.
General Trends • Alkaline metals: Increases as it goes to the right, and up the column • Transition metals: Increases as you go down the column, and meets in the middle of the row • Metalloids: Really high boiling points, generally decreases down the staircase • Nonmetals: Increases as it goes down the column and to the left
Metals • Metals have high boiling points • Alkali metals have lowest boiling points of all metals • Metallic bonding: the electrostatic attractive forces between the delocalized electrons, called conduction electrons, gathered in an "electron sea", and the positively charged metal ions • Metallic bonding in metals are strong bonds that result in high boiling point because of the higher energy needed to break the bonds between molecules
Transition metals • Transition metals generally have the higher boiling points • The middle transition metals have the highest boiling points • The reason is that they can involve the 3d electrons in the delocalization as well as the 4s. The more electrons you can involve, the stronger the attractions tend to be.
Metalloids • Very high boiling points • Up to 4000 ⁰C (Carbon) • Network covalent bonding (very strong)-bonding with covalent bonds in a network structure
Nonmetals • Low boiling points • Intermolecular forces (Van der waals forces and London-Dispersion forces) • Molecule size increases as van der waals forces increase • temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles
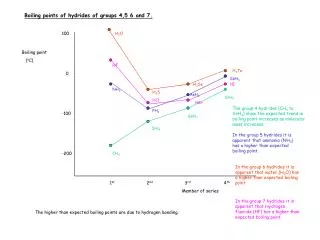
Exceptions • Metals: Mg in alkaline earth metals lower boiling point than others of the family • Nonmetals: large difference in molecule size between fluorine and other halogens caused a greater change in boiling point between fluorine and chlorine than the change between other halogens