IR spectroscopy
Adsorption of CH 2 CClF and CH 2 CBrF on TiO 2 : infrared spectroscopy and quantum-mechanical calculations Jessica Scaranto and Santi Giorgianni Università Ca’ Foscari di Venezia – Dipartimento di Chimica Fisica, Calle Larga S. Marta 2137, I-30123 Venezia, Italy.

IR spectroscopy
E N D
Presentation Transcript
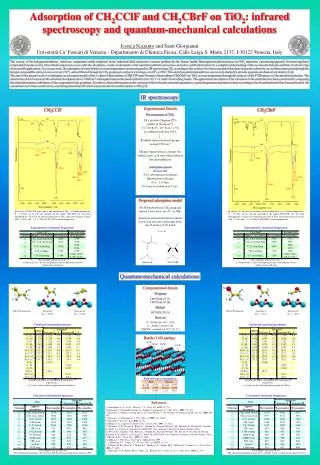
Adsorption of CH2CClF and CH2CBrF on TiO2: infrared spectroscopy and quantum-mechanical calculations Jessica Scaranto and Santi Giorgianni Università Ca’ Foscari di Venezia – Dipartimento di Chimica Fisica, Calle Larga S. Marta 2137, I-30123 Venezia, Italy The toxicity of the halogenated ethenes, which are compounds widely employed in the industrial field, represents a serious problem for the human health. Heterogeneous photocatalysis on TiO2 represents a promising approach for removing these compounds from the air [1]. Since the decomposition occurs after the adsorption, a study on the nature of the adsorbate-substrate interaction can lead to useful information for a complete understanding of the reaction mechanisms and then, for the develop of successful applications. In a recent work, the adsorption of vinyl halides at room temperature was investigated by IR spectroscopy [2]: according to the results it has been concluded that these molecules adsorb by an acid-base interaction through the halogen atom and the surface Lewis acid site (Ti4+), and an H-bond through the CH2 group and a surface Lewis basic site (O2- or OH-). This adsorbate-substrate model was successively studied by periodic quantum-mechanical calculations [3,4]. The aim of the present work is to formulate an adsorption model of the 1-chloro-1-fluoroethene (CH2CClF) and 1-bromo-1-fluoroethene (CH2CBrF) on TiO2 at room temperature through the analysis of the FTIR spectra of the adsorbed molecules. The attention has been focused on the adsorbate absorptions above 1000 cm-1 and in particular on the bands related to the C-H, C=C and C-F stretching modes. The approximate description of the vibrations of the adsorbates has been carried out by comparing the related absorptions with those of the compounds in the gas-phase. In order to obtain information on the variation of the molecular structural parameters, a periodic quantum-mechanical study according to the formulated model has been performed; the calculations have been carried out by considering the rutile (110) which represents the most stable surface of TiO2 [5]. IR spectroscopy Experimental Details Pre-treatment of TiO2 TiO2 powder (Degussa P25) [pellet of 20 mg.cm-2] T = 723 K, P ~ 10-4 Torr, t = 5 h re-oxidation with mix N2/O2 Residual surface hydroxyl groups around 3700cm-1 The pre-treated surface contains two surface Lewis acid sites which differ in the electrophilicity Adsorption spectra Background (TiO2 after the pre-treatment) Introduction of the gas (0.5 – 2.0 Torr) 20 scans at resolution of 4 cm-1 CH2CClF CH2CBrF Proposed adsorption model No H-bond between CH2 group and surface Lewis basic site (O2- or OH-) Acid-base interaction between surface Lewis acid site and a molecular basic site (F atom or C=C bond) IR spectra of CH2CClF in gas-phase and adsorbed on TiO2. (a) Room temperature, P ~ 1.0 Torr, 16 cm cell; the spectrum in the region 3800-2850 cm-1 has been multiplied by a factor of 10. Infrared spectrum of TiO2 taken after being in contact with ~ 0.6 (b) and ~ 1.2 (c) Torr of CH2CClF at room temperature. IR spectra of CH2CBrF in gas-phase and adsorbed on TiO2. (a) Room temperature, P ~ 1.0 Torr, 16 cm cell; the spectrum in the region 3800-2850 cm-1 has been multiplied by a factor of 10. Infrared spectrum of TiO2 taken after being in contact with ~ 0.6 (b) and ~ 1.2 (c) Torr of CH2CBrF at room temperature. Quantum-mechanical calculations Computational details Program CRYSTAL03 [7] CRYSTAL06 [8] Method DFT/B3LYP [9] Basis set Ti : DVAE (86-51G* ) [10] O : TVAE (8-411G) [10] CH2CFX : standard 6-31G** [11-13] Br Cl H1 H1 C1 C2 C1 C2 F H2 F H2 CH2CClF molecule Structure I Structure II Eint = -20.19 Eint = -15.41 CH2CClF molecule Structure I Structure II Eint = -18.47 Eint = -12.80 Rutile (110) surface O(2f) Ti(6f) Ti(5f) O(3f) References [1] Linsebigler, A. L.; Lu, G.; Yates Jr., J. T. Chem. Rev.1995, 95, 735. [2] Scaranto, J.; Pietropolli Charmet, A.; Stoppa, P.; Giorgianni, S. J. Mol. Struct.2005, 741, 213. [3] Scaranto, J.; Mallia, G; Giorgianni, S.; Zicovich-Wilson, C. M.; Civalleri, B.; Harrison, N. M. Surf. Sci.2006, 600, 305. [4] Scaranto, J.; Giorgianni, S. J. Phys. Chem. C2007, 111, 11039. [5] Diebold, U. Surf. Sci. Rep.2003, 48, 53. [6] Mann, D. E.; Acquista, N; Plyler, E. K. J. Chem. Phys.1955, 23, 2122. [7] Saunders, V. R.; Dovesi, R.; Roetti, C.; Orlando, R.; Zicovich-Wilson C. M.; Harrison, N. M.; Doll, K.; Civalleri, B.; Bush, I. J.; D’Arco, P.; Llunell, M. CRYSTAL03 User’s Manual, University of Torino (Torino, 2003). [8] Dovesi, R.; Saunders, V. R.; Roetti, C.; Orlando, R.; Zicovich-Wilson C. M.; Pascale, F; Civalleri, B.; Doll, K.; Harrison, N. M.; Bush, I. J.; D’Arco, P.; Llunell, M. CRYSTAL06 User’s Manual, University of Torino (Torino, 2006). [9] Becke, A.D. J. Chem. Phys.1993, 98, 5648. [10] Muscat, J. PhD Thesis, University of Manchester, 1999. [11] Hariharan, P. C.; Pople, J. A. Theoret. Chim. Acta1973, 28, 213. [12] Francl, M. M.; Petro, W. J.; Hehre W. J.; Binkley J. S.; Gordon, M. S.; DeFrees D. J.; Pople J. A. J. Chem. Phys.1982, 77, 3654. [13] Rassolov, V. A.; Ratner, M. A.; Pople, J. A.; Redfern, P. C.; Curtiss, L.A. J. Comp. Chem.2001, 22, 976.