Neonatal Hypoglycemia
1.45k likes | 4.58k Views
Neonatal Hypoglycemia. Definition. The numerical definition varies from institution to institution: Numbers based on population studies of plasma glucose concentrations during first 48-72 hours of life with hypoglycemia defined as a plasma glucose level more than 2 SD below the population mean

Neonatal Hypoglycemia
E N D
Presentation Transcript
Definition • The numerical definition varies from institution to institution: • Numbers based on population studies of plasma glucose concentrations during first 48-72 hours of life with hypoglycemia defined as a plasma glucose level more than 2 SD below the population mean • Most institutions use plasma glucose <40mg/dl on the 1st day of life and less than 40-50mg/dl after 24 hours of life • Physiologic definition: when glucose supply for cells is inadequate to meet glucose demands
Incidence • Estimated to be 10% of live births if first feeding is delayed for more than 3-6 hours after birth • Percentage is even higher for at-risk populations: • Preterm infants • SGA infants • LGA infants • Infants of diabetic mothers
Pathophysiology • Newborns have high brain-to-body weight ratio -> higher glucose demand • Impaired establishment of normal glucose homeostasis during transition from intrauterine to extrauterine life -> hypoglycemia • Normal glucose homeostasis requires supply to meet demands • Supply is dependent on adequate stores of glycogen, gluconeogenesis precursors, functioning hepatic enzymes, and a functioning endocrine system • Demands depend on the metabolic rate of the infant, which can be increased in times of stress (i.e. sepsis, asphyxia)
Clinical Manifestations • Asymptomatic • Tachypnea • Apnea • Respiratory distress • Tachycardia • Bradycardia • Jitteriness • Lethargy • Hypotonia • Weak suck • Temperature instability • Seizures
Etiology • Diminished glucose production • Increased glucose utilization from hyperinsulinemia • Increased glucose utilization without hyperinsulinemia • Metabolic Disorders • Endocrine Disorders • Other
Diminished Glucose Production • Premature infants have diminished reserves because glycogen is deposited during the 3rd trimester of pregnancy • Infants with intrauterine growth restriction (IUGR) and who are SGA have reduced glycogen stores because of: • Low intrauterine insulin levels • Chronic intrauterine hypoxia
Increased Glucose Utilization Due to Hyperinsulinemia • Infant of a diabetic mother • Maternal intrapartum treatment with glucose • Beckwith-Wiedemann syndrome • Insulinoma
Infant of a Diabetic Mother • Intermittent maternal hyperglycemia -> fetal hyperglycemia and hyperinsulinemia -> hypoglycemia once intrauterine glucose supply from mother is interrupted • Hypoglycemia occurs in 27% of infants of diabetic mothers (IDMs) • Happens in the first few hours of life • Most common in macrosomic IDMs • Premature &/or SGA IDMs are also at higher risk
Beckwith-Wiedemann Syndrome • Fetal overgrowth syndrome with characteristic features: • Macroglossia • Growth >90% • Abdominal wall defects • Ear creases/pits • Renal abnormalities • Hemi-hyperthrophy • Hyperplasia of organs (such as the pancreas)
Beckwith-Wiedemann Syndrome • Incidence: 1 in 15,000 births • Etiology: Sporadic mutation (85%), AD (15%) • 50% have transient hypoglycemia caused by hyperinsulinemia from hyperplasia of the pancreas • Increased risk for malignancy: • Wilms tumor, hepatoblastoma, neuroblastoma, gonadoblastoma • Monitored with abdominal US and alpha-fetoprotein q6months until 6 y/o
Insulinoma • Tumor of the pancreas that produces too much insulin • Very rare in children • Most are benign tumors, only about 5-10% are malignant • Treatment is surgical • If unable to surgically remove, treat with diazoxide or octreotide to reduce insulin secretion
Increased Glucose Utilization Without Hyperinsulinemia • States of stress such as hypothermia, perinatal asphyxia, sepsis, and heart failure increase usage and depletion of glycogen stores • Polycythemia - increased utilization of glucose by the increased mass of RBCs
Metabolic Disorders • Inborn errors of metabolism: • Defects in carbohydrate metabolism • Glycogen Storage Disease • Glycogen Synthase Deficiency • Galactosemia • Fructose Intolerance • Defects in amino acid metabolism • Maple Syrup Urine Disease • PropionicAcidemia • MethylmalonicAcidemia • Defects in ketogenesis and fatty acid oxidation
Endocrine Disorders • Deficiency or malfunctioning of the hormones that regulate glucose homeostasis: • Cortisol • Growth hormone • Glucagon • Epinephrine • Thyroid • These could be associated with hypothalamic, pituitary, or adrenal insufficiency
Other Causes • Maternal drugs such as terbutaline, labetalol, propranolol -> inhibit glycogenolysis and gluconeogenesis • Neurohypoglycemia: • GLUT1 transport protein facilitates glucose diffusion across blood vessels into the brain and CSF • Deficiency in GLUT1 results in low CSF glucose, but blood glucose levels are normal • Rare disorder that presents as 2-3 months with seizures, developmental delay, and acquired microcephaly
Evaluation • Blood glucose should be monitored for infants at risk for hypoglycemia: • Premature infants • SGA infants • LGA infants • IDMs • Infants whose mothers were treated with beta adrenergic agents or beta blockers • Infants under stress requiring more intensive care (i.e. sepsis, asphyxia)
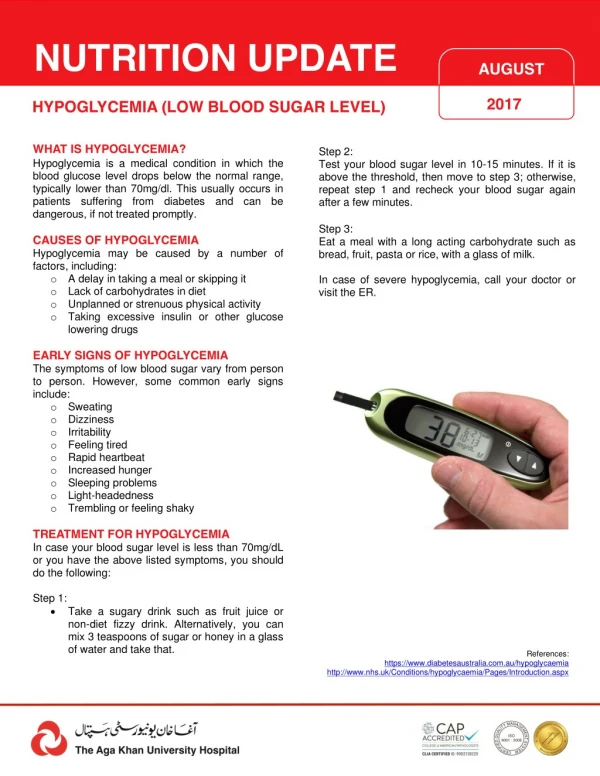
Evaluation • Monitor glucose within first 1-2 hours of life or with signs consistent with hypoglycemia • Surveillance should be continued in infants with glucose <40 until feedings well established and levels have stabilized • Low Chemstrips (glucose oxidase reagent strips for rapid screening) should be confirmed with serum glucose level processed by the lab
Evaluation • Determining Etiology: • Consider prenatal/perinatal history • Check growth parameters • Perform a careful physical exam • Screen for sepsis if suspected
Evaluation • If hypoglycemia persists for >1 week, endocrine and metabolic disorders should be suspected • Consult endocrinology • At the time of hypoglycemia, obtain: • ACTH/cortisol levels • Growth hormone levels • Insulin levels • Free fatty acids • Ketones • Pyruvate • Lactate
Evaluation • The following should also be obtained, but can be obtained at anytime: • TSH/T4 levels • Serum amino acids • Urine organic acids • Acylcarnitine profile
Management • Anticipation and prevention is key • In infants who are premature or too ill to feed, begin parenteral glucose infusion at a rate of at least 6mg/kg/min • Glucose (mg/kg/min) = (% glucose in solution x 10) x (rate of infusion per hour) / (60 x weight in kg)
Healthy asymptomatic infants • Try feeding orally with either formula, breastmilk, or D10W • Use of formula or breastmilk better than D10W because they provide carbohydrates as well as protein and fats that are metabolized more slowly to provide a sustained supply of substrate • Recheck glucose in 20-30 mins after the feeding and continue to feed q2-3 hrs • Blood glucose should be followed before each feed for 12-24 hours
Symptomatic infants or infants with very low glucose concentrations • Start parenteral glucose infusions on: • Symptomatic infants • Infants with a glucose of <20-25 • Infants who do not tolerate enteral feedings • Infants whose blood sugar remains <40 after a trial of oral feeding
Symptomatic infants or infants with very low glucose concentrations: • Start with a bolus of 2-4ml/kg of D10W • Then begin a glucose infusion of at least 6mg/kg/min • Check blood glucose 20-30 mins after bolus to determine if another bolus is needed, and adjust rate of dextrose concentration to maintain plasma glucose >45mg/dl • Follow blood glucose every 1-2 hours until stable, then can space out monitoring as needed • When the glucose concentration is stable for 12-24 hrs, the glucose infusion rate can be tapered slowly by 10-20% each time the feeds are advanced and the pre-prandial blood glucose is >50-60 mg/dl
Persistent Hypoglycemia (> 7 days) • Corticosteroids stimulate gluconeogenesis and reduces peripheral glucose utilization • should be considered in infants who remain hypoglycemic after 2-3 days of glucose infusion of >12mg/kg/min • Glucagon can also be used during severe hypoglycemia as a temporizing measure in infants with adequate glycogen stores (i.e. NOTin SGA or premature infants) • Diazoxide/Somatostatin/Octreotide inhibits insulin release for those with persistent hypoglycemia and hyperinsulinemia
Persistent Hypoglycemia (> 7 days) • Human growth hormone for infants with growth hormone deficiency • Nifedipine – case reports have shown some success with few side effects • Subtotal pancreatectomy for hyperinsulinemia • hypoglycemia recurs in up to 1/3 of patients • 40-60% develop DM later in life
Prognosis • Symptomatic hypoglycemia can result in brain injury • Most common sequelae are: • Disturbances in neurological development and intellectual function • Motor deficits (spasticity and ataxia) • Seizures * May be related to the underlying etiology of the hypoglycemia • There is inconclusive evidence on the effect of asymptomatic hypoglycemia on neurodevelopment