proteins
proteins. proteins. Proteins are polymers composed of sub-units called amino acids that are linked by peptide (amide) bond. Amino acid. amino acid are molecules containing an amine group, a carboxylic acid group and a side-chain that varies between different amino acids. Amino acid.

proteins
E N D
Presentation Transcript
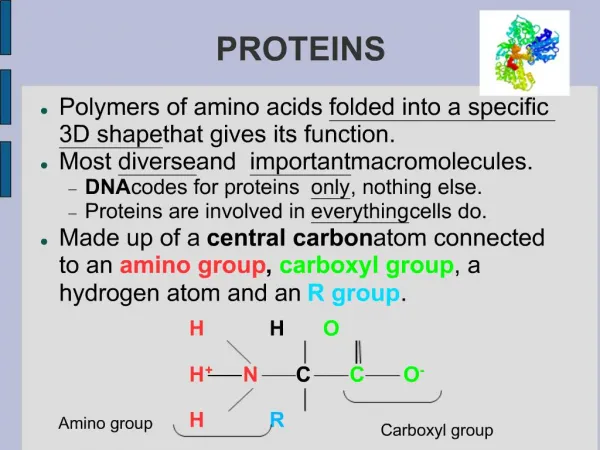
proteins • Proteins are polymers composed of sub-units called amino acids that are linked by peptide (amide) bond.
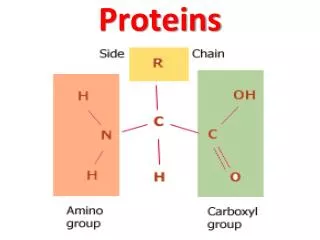
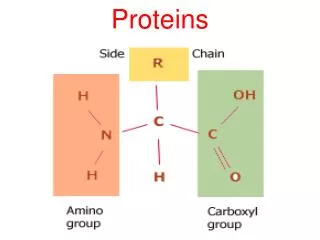
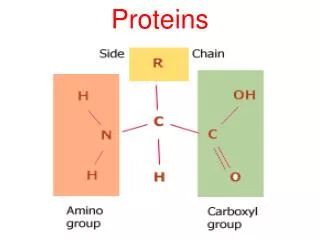
Amino acid • amino acid are molecules containing an amine group, a carboxylic acid group and a side-chain that varies between different amino acids.
Amino acid • general formula for α-amino acid is where R is an organic substituent. The amino group is attached to the carbon atom immediately adjacent to the carboxylate group (the α–carbon ).
Amino acid: There are two types of amino acids: • non-essential amino acids can be made by the body. • essential amino acids cannot be made by the body and must be taken from food. There are 8 essential amino acids.
Amino acid • essential amino acid isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. • histidine is considered semi-essential because the body does not always require dietary sources of it.
Importance of proteins: • Structural Proteins (Membranes, cartilages, Connective tissues). • Transport Proteins • Direct biochemical reactions (enzymes) • Defend the body against infection (antibodies) • Control metabolic process (hormones) • Contractile Proteins - are responsible for movement
albumin Albumin is the main protein of blood plasma; it binds to water, cations (such as Ca2+, Na+ and K+), fatty acids, hormones, bilirubin and drugs
gelatin • Gelatin is a translucent, colorless, brittle (when dry), tasteless solid substance • derived from the collagen inside animals' skin and bones. • It is commonly used as a gelling agent in food, pharmaceuticals, and cosmetic manufacturing.
a. Protein solubility • All proteins dissolve in dilute acid ,base and water
b. Coagulation and precipitation test 1.precipitation by heat: White turbidity will appear
b. Coagulation and precipitation test 2.precipitation by conc. Acid: • Add 2ml of conc. Nitric acid in a test tube then in the wall of the tube add 2ml of protein solution • Result White ppt will appear
b .Coagulation and precipitation test 3.Precipitation by minerals • Result: Ppt will appear
b. Coagulation and precipitation test 4.precipitation by alkaloids • Result: Ppt will appear
b. Coagulation and precipitation test 5.preciptation by alcohol : Add to 3ml protein solution 6ml of(70-90%) of ethyl alcohol • Result: Ppt will appear
c. Gel formation test: • Dissolve 0.5 g of gelatin in 10ml hot H2O in test tube then cool the test tube in ice bath • Result: • Gel will formed
Chemical tests: General test for all protein • Biuret test: Add 3ml of protein solution , 3ml of 10% NaOH with shake, then add 2 drops of 1% CuSO4 after shake violet color will appear
Chemical tests Differentiation(specific) test: • Millon test: Add 2ml of protein solution, 5drops of millon’s reagent. Result: +ve albumin (white ppt) -ve gelatin (no white ppt) Then heat the test tube . red ppt will appear with (albumin and gelatin)
Chemical tests: Differentiation (specific) test: • Lead acetate test : Add to 0.5 ml of protein solution 0.5ml of 20% NaOH heat the test tube in water bath for 1 min. then add 2 drops of conc.lead acetate Result: Brown color(+ve) with albumin -ve with gelatin