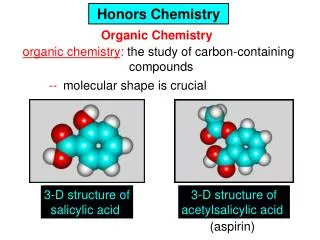
Organic chemistry
480 likes | 847 Views
Organic chemistry. Organic Compounds. Organic Compounds - any covalently bonded compound containing carbon except carbonates CO 3 - , carbon monoxide CO, carbon dioxide CO 2 and acetates C 2 H 3 O 2 -. A. Organic Nomenclature. General Characteristics of Organic Molecules.

Organic chemistry
E N D
Presentation Transcript
Organic Compounds • Organic Compounds - any covalently bonded compound containing carbon • except carbonates CO3-, carbon monoxide CO, carbon dioxide CO2 andacetates C2H3O2-
General Characteristics of Organic Molecules • Carbon always forms 4 covalent bonds • due to its hybridization of its s and p • orbitals
Stabilities of Organic Molecules • Carbon forms very strong bonds between H, O, N, and halogens. • Carbon also forms strong bonds with itself. • Therefore, C can form stable long chain or ring • structures. • Bond strength increases from single to double to • triple bond. • Bond length decreases in the same direction.
Hydrocarbons • Hydrocarbons- Organic compounds that contain only carbon & hydrogen • Alkanes- contain only single covalent bonds • Alkenes- contain one or more carbon - carbon • double bond • Alkynes- contain one or more carbon-carbon triple • bond
Saturated & Unsaturated Hydrocarbons • Saturated hydrocarbons – contain only single carbon-carbon bonds (alkanes) • Unsaturated hydrocarbons – contain double • carbon-carbon bonds (alkenes) • or triple carbon-carbon (alkynes) bonds
Formulas • Alkanes = CnH2n+2 • Alkenes = CnH2n • Alkynes = CnH2n-2
Nomenclature • Must memorize prefixes • To name, look at the formula for the hydrocarbon • Determine if it is an alkane, alkene, or alkyne • Use the prefix for the number of carbons • Add ending (ane, ene, yne)
Structural Formulas • “Lazy” way to write the Hydrogens • Instead of drawing the bonds, just state how many hydrogens are attached • NOTE: The bonds are between CARBONS in a parent chain, and not hydrogens! Structural Formula Lewis Structure
B. the first ten alkanes • CH4 - methane • C2H6 - ethane • C3H8 - propane • C4Hl0 - butane • C5H12- pentane
C6H14 - hexane • C7H16 - heptane • C8H18 - octane • C9H20 - nonane C10H22 - decane
C. the general formula is CnH 2n +2 1. where n stands for the number of carbons D. Branched chain saturated hydrocarbons 1. must name the longest straight chain 2. number the straight chain so the branches have the lowest number
3 list the branches in alphabetical order e.g. 3-ethyl 2 methyl hexane
e.g. 3-ethyl 2 methyl hexane Draw back bone for compound = in this case it is hexane C C-C-C-C-C-C 1 2 3 4 5 6 C C Number the carbons Attach branch group Make sure all C have 4 bonds
Designate the Location Designate the location (number of the carbon on the parent chain) for each attached group 2-methyl 1 2 3 4 5 2 - methyl pentane
Some Simple Alkanes 2-methylpentane • 2,2-dimethylbutane 3-ethylhexane • 2,3-dimethylbutane
E. homologous series - differs from its neighbors by only methylene group (CH2)
II. Types of formulas Structural formulas - shows the structure of a molecule B. Condensed structural formulas
III. cyclic hydrocarbon • A. named with the prefix cyclo • e.g. cyclohexane
Drawing and Naming Cycloalkanes Cycloalkanes are represented by polygons. A triangle represents cyclopropane, a square represents cyclobutane, a pentagon represents cyclopentane, and so on.
Alkenes and Alkynes • Unsaturated • contain carbon-carbon double and triple bond to which more hydrogen atoms can be added. • Alkenes: carbon-carbon double bonds • Alkynes: carbon-carbon triple bonds.
IV. alkenes - doubled bonded hydrocarbons considered to be unsaturated a. the double bond is located between two C i. ethene • H- C = C - H • I I • H H b. change the ane ending to ene
c. in very large molecules you must give a number where the double bond begins e.g. 2- butene d. If there are more than one double bond the ene is preceded by number of double bonds 1.3 pentadiene V. Alkynes - triple bonded hydrocarbons - also unsaturated A. the triple bond is located between the two C 1. ethyne H-C=C-H
B. change the ane ending to yne C. must also indicate where the triple bond starts e.g. 2-pentyne
Naming Alkenes and Alkynes When the carbon chain has 4 or more C atoms, number the chain to give the lowest number to the double or triple bond. 1 2 3 4 CH2=CHCH2CH3 1-butene CH3CH=CHCH3 2-butene CH3CCCH3 2-butyne
Endings • Alkanes (all C-C single bonded parent chain) end in –ane • MethaneCH4 • EthaneC2H6 • PropaneC3H8 • Attached carbon groups (substituents) end in –yl • Methyl CH3 - • EthylCH3CH2- • Propyl CH3CH2CH2 – 3-ethylpentane
VII alkyl halides The attachment of a Halogen to a carbon group (F, Cl, Br, I) Chloro methane dicloro, difluoro methane 2 – bromo butane
VII. alcohols - contain OH groups A. ethanol B. change the e to ol C. methane to methanol VIIIKetones - contains double bonded oxygen located anywhere but the end A. change to e to one B. propane to propanone
IXaldehydes - contains double bonded oxygen only at the end A. change e to al B. ethane to ethanal X carboxylic Acid - contain a double bonded oxygen and a hydroxylgroup A. change to e to oic acid B. ethane to ethanoic acid X I ethers A. two alkyl groups attached to an oxygen B. dimethyl ether
Aromatic Compounds and Benzene Aromatic compounds contain benzene. Benzene, C6H6 , is represented as a six carbon ring with 3 double bonds. Two possible resonance structures can be drawn to show benzene in this form.
When Benzene rings are attached to groups that have higher priorities – it is known as a phenyl group (C6H5-)
CO2H O I N C R E A S I N G -CH O C -OH C=C R-,C6H5-,Cl-, Br-, NO2
Isomers • Straight chain alkanes: An alkane that has all its carbons connected in a row. Branched chain alkanes: An alkane that has a branching connection of carbons. • Isomers:Compounds with same molecular formula • but different structures.
F. structural isomers 1. same molecular formula put differ in structural formula 2. 4 carbon alkane has two possible isomers 3. C5H12 has 3 isomers 4. C6H14 has 5 isomers 5. C10H22has 75
Carbons in butane (C4H10) can be arranged in two ways; four carbons in a row (linear alkane) or a branching (branched alkane). These two structures are two isomers for butane.
Different isomers are completely different compounds. They have different structures, different physical properties such as melting point and boiling point, and may have different physiological properties.
Learning Check • Draw all possible structural isomers of C5H12 pentane 2-methlyl butane 2,2 – dimethyl propane
Example • Name C3H8 • This falls under the equation CnH2n+2 • Therefore it is an alkane • Since n=3 we will use the prefix prop • Since it is an alkane we will use the ending ane • propane
Example • What is the formula for octene? • Since the ending is -ene we know that it is an alkene (CnH2n) • Since the prefix is oct we know there are 8 carbons • C8H16
Example • Name C5H12 • 5 carbons = pent • CnH2n+2 = alkane (ane) • pentane