Enzyme Structure and Function
Enzyme Structure and Function. Protein catalysts. Enzymes are Catalysts. This means that enzymes help speed up chemical reactions. How? Enzymes lower the activation energy of a reaction.

Enzyme Structure and Function
E N D
Presentation Transcript
Enzyme Structure and Function Protein catalysts
Enzymes are Catalysts • This means that enzymes help speed up chemical reactions. • How? • Enzymes lower the activation energy of a reaction. • If the Ea is lower, more reactant molecules will make it over the “energy hill”, so more products are produced per second.
Analogy: Plasma HD TVs • What has happened to the rate of purchase of plasma HD TVs over the last 10 years? • The rate has increased (i.e.: Best Buy might have sold an average of 2 per month in 1997 but sell an average of 25 per month in 2007) • Just like, with enzymes, the rate of the reaction increases. • Why has the number changed? • The price has decreased, allowing more people to overcome the “cost barrier”. • Enzymes lower the “energy barrier” so more reactants can overcome it.
Lock and Key Model • Enzymes are specific • each one helps only one type of reaction to occur. • Each enzyme has a unique “active site”. • An enzyme binds a substrate molecule in its active site. • When the substrate is bound to the enzyme, the bonds are easier to break, so less energy is required for the reaction to occur.
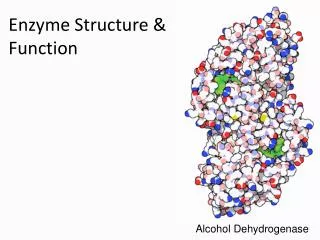
Enzymes are Organic Catalysts • Organic compound = made by living things, carbon based. • Lipids, carbohydrates, proteins, nucleic acids • Enzymes are proteins. • Proteins are polypeptides. They are long chains of amino acids. • The number, type, and sequence of amino acids determines a protein’s shape. • So, what makes each enzyme different (i.e.: the active sites different) are the amino acids that make up each enzyme.
“Magnified” section of protein (unfolded) This is the enzyme “catalase”, the enzyme found in liver Enzyme (and protein) structure
Amino Acids • “Building Blocks” of proteins. • 20 different ones • Each one has an amino group, a carboxyl group, and an “R-group”. • The R-group of each amino acid is unique.
General Structure Specific Examples
DNA and Proteins • The N-base sequence (ATCG) of a gene is the “recipe” for making a protein. • The N-base sequence dictates the correct sequence of amino acids.
More on Protein Structure • An enzyme (and any other protein) is made by linking amino acids together in the correct order (sequence). • Reaction = dehydration synthesis (condensation) reaction. • Many of these reactions occur until the protein consists of 100+ amino acids linked together. • http://trc.ucdavis.edu/biosci10v/bis10v/media/ch02/peptide_bonding.html
Your Turn! • Draw a dipeptide that consists of serine and threonine.
The amino acid chain (polypeptide) twists and folds to form a functional protein • The sequence of amino acids making up an enzyme determines how that enzyme folds into the correct 3-D shape, allowing it to bind with its substrate. • So a “real” enzyme looks something like this.
An enzyme cannot function if it has an improper shape • Environmental factors: • May temporarily change the shape of the enzyme = denaturation • May “block” the active site, rather than change the shape • Genetic disorder: • Enzyme never folds into the correct shape because the “recipe” is wrong. • Adding the wrong amino acid(s) is like adding the wrong ingredient(s) = incorrect product.
The Story so far… • Substrate Concentration: • Decreases over time • Less collisions between substrate & enzyme = decreased reaction rate • Bubbling slows due to less hydrogen peroxide molecules colliding with liver enzymes. (Remember the toothpicks) • When all of the substrate has been used, the reaction stops.
Heavy Metal Concentration: • As heavy metal concentration increases, reaction rate decreases. • The copper sulfate soaked liver had a reaction rate of 1 vs. the water soaked liver had a reaction rate of 3. • A heavy metal can bind to an enzyme’s active site. • Think of the straws mixed with toothpicks. • Enzyme can’t bind to substrate as often so the reaction is slower.
Metal Ions and Enzyme Inhibition Hydrogen peroxide (substrate) binds to enzyme and is broken down (reaction occurs-bubbling observed.) Metal ion gets in the way and hydrogen peroxide cannot bind to the enzyme. Reaction either doesn’t occur or occurs at a slower rate. (Not all enzymes are blocked.)
pH • For catalase (liver enzyme) a neutral pH (7) is ideal. Lowering or raising pH decreases reaction rate. • Acid (pH of 2): reaction rate of 1 • Base (pH of 12): reaction rate of 2 • H+ and OH- ions do NOT block the enzyme’s active site. They DO change the 3-D shape of an enzyme, thereby changing its active site. • “Lock and Key” no longer match; enzyme can’t bind to substrate when they collide.
pH and denaturation Not all enzymes have the same “optimal” pH. Catalase (liver enzyme) is more like chymotrypsin. However, pepsin (a stomach enzyme) functions best at a low (acidic) pH. At pH 1, pepsin is in it’s functional shape; it would be able to bind to its substate. At pH 5, the enzyme’s shape is different and it no longer has an active site able to bind the substrate. The change in enzyme activity is observed as a difference in reaction rate.
Temperature • The ideal temperature for the functioning of catalase is around 40°C (human body temp.) • At lower temperatures enzyme and substrate molecules move less, so fewer collisions occur and the reaction is slower • At higher temperatures the 3-D shape of the enzyme changes and the active site takes on a different shape (denaturation) • Substrate can’t fit into active site
Temperature and Reaction Rate Shape of active site changes (substrate can’t bind) Slower reaction due to fewer collisions