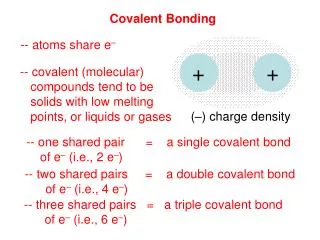
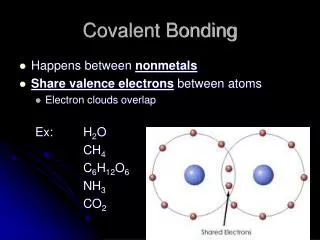
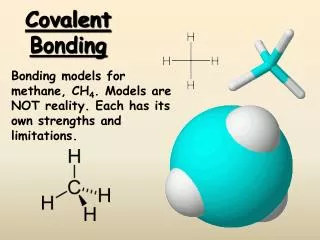
Covalent Bonding
Covalent Bonding. P 104 in text book Use full name for first element and add “ ide ” to second (just like you do for ionic formulas) BUT…unlike for ionic formulas… Prefixes are used to denote how many of each atom the prefix is in front of. Naming of covalent compounds Non-metals. mono = 1

Covalent Bonding
E N D
Presentation Transcript
P 104 in text book • Use full name for first element and • add “ide” to second (just like you do for ionic formulas) • BUT…unlike for ionic formulas… • Prefixes are used to denote how many of each atom the prefix is in front of Naming of covalent compoundsNon-metals
mono = 1 • di = 2, ex: CO2 = carbon dioxide • Notice in above example that “mono” is not used in front of the carbon. “Mono” is implied in front of the first element. • Tri = 3, ex: BF3 = boron trifluoride Naming Covalent Compounds
tetra = 4 • penta = 5 • hexa = 6 • hepta = 7 • octa = 8 • try naming: N2O5 • dinitrogenpentoxide • notice that we drop the “a” ending of penta when the next word starts with a vowel. • This is the case for all endings with vowels, except di, as in dioxide, and tri, as in trioxide. Naming Covalent Compounds
If three elements combine to share electrons, the shapes of the s orbital and p orbitals of the central atom change. • We call the new shape an sp1 hybrid • recall, the original s orbital is a spherical shape • the original p orbital is a sort of dumbell shape. Hybridization of Central Atom
the new sp1 hybrid looks like a weighted dumbell, • the electrons are being pulled toward the other atom they are being shared with. Hybridization of Central Atom
to make a new shared linear shape – see your hand-out Hybridization of Central Atom
If central atom shares with three other atoms, • it creates an sp2 orbital hybrid, • because it involves changing the shapes of the central atom’s s-orbital • and 2 of its p-orbitals. • the new compound shape is trigonal planar. Hybridization of Central Atom
If the central atom shares with four other atoms, or has a free pair of electrons, we get an sp3 hybrid, • because the s orbital and all 3 p orbitals shapes are hybridized. • This creates a tetrahedral shape of the molecule. Hybridization of Central Atom
Text reference: p 113 – 114 • Acids – substances that release H+ ions (protons) when dissolved in water. • First element is always hydrogen • 2 types: • contain oxygen or • don’t contain oxygen Naming Acids
Prefix “hydro” • Suffix “ic” • “Acid” at end • Ex: HCl = hydrochloric acid • You try: HCN • hydrocyanic acid • H2S • hydrosulfuric acid Naming Acids not Containing Oxygen
2 types of oxyanions: • oxyanion ends in “ate” • ex: sulfate, fluorate • oxyanion ends in “ite” • ex: sulfite, fluorite Naming Acids whose anions contain oxygen (oxyanions)
Acids whose oxyanions end with “ate”: • use root name of oxyanion + “ic” • then add “acid” • think – “I ate that acid … ic!” • H2SO4 • SO42- = sulfate • sulfuric acid • you try: H3PO4 • phosphoric acid • HC2H3O2 • acetic acid Naming Acids With “ate” Oxyanions
Acids whose oxyanions end with “ite”: • use root name of oxyanion + “ous” • then add “acid” • ex: H2SO3 • SO32- = sulfite • sulfurous acid • You try: HNO2 • nitrous acid • HClO • hypochlorous acid Naming Acids With “ite” Oxyanions
Some elements are never found alone. • They hang out in pairs if they’re not bonded to something else. • Oxygen is an example. • When we refer to oxygen, unless we’re being more specific, we mean O2. • The others are the elements contained in the BrINClHOF family name. • Can you guess them? The BrINClHOF family
Bromine • Iodine • Nitrogen • Chlorine • Hydrogen • Oxygen • Fluorine Basically group 17 + hydrogen, oxygen and nitrogen. The BrINClHOF family