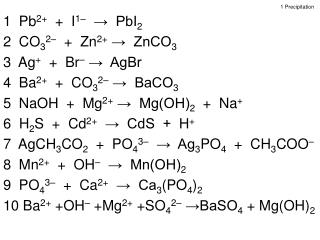1 Precipitation
1 Precipitation. 1 Pb 2+ + I 1 – → PbI 2 2 CO 3 2– + Zn 2+ → ZnCO 3 3 Ag + + Br – → AgBr 4 Ba 2+ + CO 3 2– → BaCO 3 5 NaOH + Mg 2+ → Mg(OH) 2 + Na + 6 H 2 S + Cd 2+ → CdS + H + 7 AgCH 3 CO 2 + PO 4 3– → Ag 3 PO 4 + CH 3 COO –

1 Precipitation
E N D
Presentation Transcript
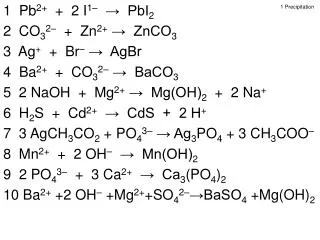
1 Precipitation 1 Pb2+ + I1– → PbI2 2 CO32– + Zn2+ → ZnCO3 3 Ag+ + Br– → AgBr 4 Ba2+ + CO32– → BaCO3 5 NaOH + Mg2+ → Mg(OH)2 + Na+ 6 H2S + Cd2+ → CdS + H+ 7 AgCH3CO2 + PO43– → Ag3PO4 + CH3COO– 8 Mn2+ + OH– → Mn(OH)2 9 PO43– + Ca2+ → Ca3(PO4)2 10 Ba2+ +OH– +Mg2+ +SO42– →BaSO4 + Mg(OH)2
2 Acid/Base 1 NaCN + H2O → HCN + Na+ + OH– 2 CH3COOH + HCO3– → H2O + CO2 + CH3COO– 3 CO32– + H+→ HCO3– 4 CH3COOH + MgCO3 → Mg2+ + Ac– + H2O+CO2 5 Zn(OH)2 + OH–→ Zn(OH)42– 6 F– + H+→ HF 7 H+ + Cu(OH)2→ Cu2+ + H2O 8 OH– + NH4Cl → NH3 + H2O + Cl– 9 OH– + H3PO4→ H2O + HPO42– 10 H+ + NO2–→ HNO2
3 Redox 1 H+ + MnO4– + Sn2+ → Mn2+ + Sn4+ + H2O 2 H+ + I– + IO3– → I2 + H2O 3 H+ + MnO4– + Cl– → Mn2+ + Cl2 + H2O 4 Br2 + I– → I2 + Br– 5 Sn2+ + Fe3+→ Sn4+ + Fe2+ 6 H+ + MnO2 + Br– → Mn2+ + Br2 + H2O 7 Cr2O72– + SO32– → Cr3+ + SO42– 8 H2O2 + I– → H2O + I2 9 H+ + Fe2+ + MnO4– → Fe3+ + Mn2+ + H2O 10 H+ + Na2Cr2O7 + Br– → Na+ +Br2 + Cr3+ +H2O
4 Metal Displacement 1 Zn + Sn2+→ Sn + Zn2+ 2 Sn2+ + Ag+→ Sn4+ + Ag 3 Zn + Cu2+→ Cu + Zn2+ 4 Cl2 + Br– → Cl– + Br2 5 Ca + H+→ Ca2+ + H2 6 Pb + Ag+→ Pb4+ + Ag 7 Na + H2O → Na+ + OH– + H2 8 Fe + Fe3+→ Fe2+ 9 Al + Fe2+→ Al3+ + Fe 10 MnO4– + SO32– → MnO2 + SO42–
5 Types 1-4 1 AgCH3CO2 + CO32– → Ag2CO3 + CH3COO– 2 NaCN + H2O → HCN + Na+ + OH– 3 Al + Ni2+→ Ni + Al3+ 4 OH– + Mg2+ → Mg(OH)2 5 Cs + H2O → Cs+ + OH– + H2 6 H+ + SO42– + Ba2+ + Ac– → BaSO4 + CH3CO2H 7 Zn + Cu2+→ Cu + Zn2+ 8 NH4+ + OH– → NH3 + H2O 9 H+ + SO42– + CaF2 → CaSO4 + HF 10 H2S + Ni2+→ H+ + NiS
5 Types 1-4 11 Zn2+ + PO43– → Zn3(PO4)2 12 CaCO3 + H+→ H2O + CO2 + Ca2+ 13 I– + Pb2+ → PbI2 14 OH– + CH3CH2COOH→ H2O + CH3CH2COO– 15 BaO + H2O → Ba(OH)2
6 Types 1-4 1 Fe3+ + I– → Fe + I2 2 H2S + Ag+→ H+ + Ag2S 3 MnO4– + H+ + Cl– → Mn2+ + Cl2 + H2O 4 Sn(CH3CO2)2 + PO43– → Sn3(PO4)2 + Ac– 5 NaF + H2O → HF + Na+ + OH– 6 Cu2+ + OH– → Cu(OH)2 7 CO32– + H+ → HCO3– 8 H+ + ZnS → Zn2+ + H2S 9 Cr2O72– + SO32– → Cr3+ + SO42– 10 OH– + H3PO4 → H2O + HPO42–
6 Types 1-4 11 Zn + Cu2+→ Zn2+ + Cu 12 MnO4– + H2SO3 → SO42– + Mn2+ + H2O 13 PH3 + BCl3 → H3PBCl3 14 CH3CO2H +Ba(OH)2→ Ba2+ + CH3CO2– +H2O 15 Ag+ + Fe2+ → Ag + Fe3+
7 Types 1 – 7 1 C6H5Cl + O2→ CO2 + H2O + HCl 2 CO2 + OH– → CO32– + H2O 3 HNO2 + OH– → H2O + NO2– 4 Cu(OH)2 + NH3→ Cu(NH3)42+ + OH– 5 OH– + Mg2+ → Mg(OH)2 6 Ag2S + H+→ Ag+ + H2S 7 SO2 + OH–→ SO32– + H2O 8 N2O3 + H2O → HNO2 9 NH3 + Co2+→ Co(NH3)42+ 10 C2H5OH + O2→ CO2 + H2O
7 Types 1 – 7 11 Pb2+ + I–→ PbI2 12 SCN– + Fe3+ → Fe(SCN)2+ or Fe(SCN)63– 13 Na2SO3 + H2O → Na+ + H2SO3 + OH– 14 AgCl + CN–→ Ag(CN)2– + Cl– 15 Li2O + H2O → Li+ + OH–
8 Types 1 – 10balanced! 1 Ca + 2 H+→ Ca2+ + H2 2 H+ + SO42– + Ba2+ + OH– → H2O + BaSO4 3 14H+ +3Sn2+ + Cr2O72–→ 2Cr3+ + 3Sn4+ + 7H2O 4 Br2 + 2 I– → I2 + 2 Br– 5 2 CN– + Ag+ → Ag(CN)2– 6 2 H2O → 2 H2 + O2 7 2 H+ + CaO → Ca2+ + H2O 8 NaF + H2O → Na+ + OH– + HF 9 CO2 + Ca2+ + 2 OH– → CaCO3 + H2O 10 2 H+ + SO32– → H2SO3
8 Types 1 – 10balanced 11 Ag+ + Br– → AgBr 12 2 KClO3→ 2 KCl + 3 O2 13 3 Cl2 + 2 Fe → 2 FeCl3 14 BH3 + NH3→ H3NBH3 15 Zn(NO3)2 + 4 OH– → Zn(OH)42– + 2 NO3–
9 Types 1 – 10 balanced 1 Ni + Cu2+→ Ni2+ + Cu 2 2 Ag+ + CrO42–→ Ag2CrO4 3 H2 + CuO → Cu + H2O 4 NH3 + CH3COOH → CH3COO– + NH4+ 5 (NH4)2CO3+Ba2++2 OH–→BaCO3+ 2NH3+ 2H2O 6 N2O3 + H2O → 2 HNO2 7 H2O +2MnO4–+3SO32–→2MnO2+3SO42–+2 OH– 8 Al3+ + 6 OH– → Al(OH)63– 9 Zn2+ + CrO42–→ ZnCrO4 10 H2O2 + 2 Br– + 2 H+→ 2 H2O + Br2
9 Types 1 – 10 balanced 11 3 Mg + N2→ Mg3N2 12 Ni2+ + S2–→ NiS 13 2 Cu2+ + 2 H2O → 2 Cu + O2 + 4 H+ 14 Cl2 + 2 OH–→ OCl– + H2O + Cl– 15 Mg3N2 + 3 D2O → 3 MgO + 2 ND3
10 1 Ba2+ + SO42– → BaSO4 2 2 CH3COOH+CO32–→H2O+ CO2 + 2 CH3COO– 3 Pb(NO3)2 + 2 Cl–→ PbCl2 + 2 NO3– 4 16H+ +2MnO4– + 5Sn2+→5Sn4+ + 2Mn2+ + 8H2O 5 Na + 2 H2O → Na+ + 2 OH– + H2 6 Cu2+ + 2 OH–→ Cu(OH)2 7 H+ + CO32–→ HCO3– 8 Fe3+ + H3PO4→ FePO4 + 3 H+ 9 SO2 + H2O2→ 2 H+ + SO42– 10 2 Fe + 3 Cu2+→ 2 Fe3+ + 3 Cu
10 11 NaH + H2O → H2 + Na+ + OH– 12 14H+ + Cr2O72– + 6Br– → 3Br2 + 2Cr3+ + 7H2O 13 F– + H+→ HF 14 2 H+ + CaO → Ca2+ + H2O 15 Cl2 + 2 Br–→ 2 Cl– + Br2
11 1 Ti + O2→ TiO2 2 SO2 + CaO → CaSO3 3 Pb + 4 Ag+→ Pb4+ + 4 Ag 4 3 Mg + N2→ Mg3N2 5 CH3COOH + HCO3–→CH3COO– + H2O + CO2 6 CuO + 2 H+→ Cu2+ + H2O 7 2 H2O2→ 2 H2O + O2 8 2 PbS + 3 O2→ 2 PbO + 2 SO2 9 2 H+ + SO42- + Ca(CN)2→ 2 HCN + CaSO4 10 2 KClO3→ 2 KCl + 3 O2
11 11 2 V3+ + 3 CO32–→ V2(CO3)3 12 4 NH3 + Zn2+→ Zn(NH3)42+ 13 2 AgNO3 + CrO42–→ Ag2CrO4 + 2 NO3– 14 CaC2 + 2 H2O → Ca(OH)2 + C2H2 15 2 Al + 6 OH– + 6 H2O → 2 Al(OH)63– + 3 H2
12 2000 a Ca + 2 H2O → Ca(OH)2 + H2 b C4H9OH + 6 O2→ 4 CO2 + 5 H2O c 4 NH3 + Ni2+→ Ni(NH3)42+ d Cu2+ + S2– → CuS e Sn2+ + 2 Ag+→ Sn4+ + 2 Ag f SrO + H2O → Sr(OH)2 h 3 CO + Fe2O3→ 2 Fe + 3 CO2
12 1998 a Sn2+ + 2 Fe3+→ 2 Fe2+ + Sn4+ b Co2+ + 2 OH–→ Co(OH)2 c C2H4 + 3 O2→ 2 CO2 + 2 H2O d H3PO4 + OH–→ H2O + H2PO4– e CaSO3→ CaO + SO2 f 2 H+ + Cl– + Ag(NH3)2+→ AgCl + 2 NH4+ g Na2O + H2O → 2 Na+ + 2 OH– h Zn + 2 H+→ Zn2+ + H2
13 1997 a 6 OH– + Al3+→ Al(OH)63– b 6 H+ + 5 Br– + BrO3–→ 3 Br2 + 3 H2O c SO2 + H2O → H2SO3 d PH3 + BCl3→ H3P-BCl3 e H2 + FeO → Fe + H2O f KNH2 + H2O → NH3 + K+ + OH– g 3 Mg + N2→ Mg3N2 h Ni2+ + S2–→ NiS
13 1995 a C2H5OH + 3 O2→ 2 CO2 + 3 H2O b BaO + H2O → Ba2+ + 2 OH– c Cl2 + 2 OH–→ H2O + OCl– + Cl– d 4 H+ + 4 Fe2+ + O2→ 4 Fe3+ + 2 H2O e 6H+ + 3SO42– +Ca3(PO4)2→2H3PO4 + 3 CaSO4 f H2S + Hg2+→ HgS + 2 H+ g CaH2 + 2 H2O → 2 H2 + Ca2+ + 2 OH– h Zn + Cu2+→ Cu + Zn2+
14 2001 a SO2 + H2O → H2SO3 b SCN– + Fe3+→ Fe(SCN)2+ c Cu + 2 Ag+→ 2 Ag + Cu2+ d OH– + CH3CH2COOH → H2O + CH3CH2COO– e 14H+ + 6Fe2+ + Cr2O72–→2Cr3+ + 6Fe3+ + 7H2O f Cl2 + 2 Br–→ 2 Cl– + Br2 g Sr2+ + SO42–→ SrSO4 h MgCO3→ MgO + CO2
14 2002 a 2 I– + Pb2+→ PbI2 b P4 + 5 O2→ P4O10 c Cs2O + H2O → 2 Cs+ + 2 OH– d 4 Cl– + Co2+→ CoCl42– e 2 NaHCO3→ Na2CO3 + H2O + CO2 f 2 H+ + ZnS → Zn2+ + H2S g 8 H+ + MnO4– + 5 Fe2+→5 Fe3+ + Mn2+ + 4H2O h OH– + NH4Cl → NH3 + H2O + Cl–
15 2002 Form B a 2 C3H7OH + 9 O2→ 6 CO2 + 8 H2O b CrO42– + Pb2+→ PbCrO4 c Fe + 2 Fe3+→ 3 Fe2+ d 4 NH3 + Cu2+→ Cu(NH3)42+ e SO2 + H2O → H2SO3 f PO43– + H+ → HPO4– g HCl + CN– → HCN + Cl– h Br2 + 2 I– → 2 Br– + I2
15 2005 Form B a CO32– + Sr2+ → SrCO3 b 2 C3H6 + 9 O2→ 6 CO2 + 6 H2O c 4 NH3 + Zn2+→ Zn(NH3)42+ d CH3COOH + OH–→ CH3COO– + H2O e 2 K + 2 H2O → 2 K+ + 2 OH– + H2 f Fe + S → FeS g H+ + F–→ HF h Pb + 4 Ag+ → Pb4+ + 4 Ag