Elements, Compounds, Mixtures
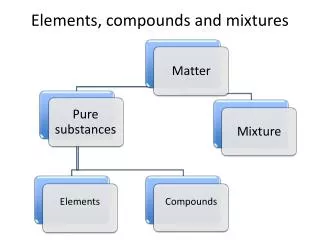
Elements, Compounds, Mixtures. atom. The smallest part into which an element can be divided and still maintain its properties. element. Any substance that cannot be broken down into a simpler substance. compound. A substance that is formed by the combination of elements using fixed amounts.

Elements, Compounds, Mixtures
E N D
Presentation Transcript
atom The smallest part into which an element can be divided and still maintain its properties
element Any substance that cannot be broken down into a simpler substance
compound A substance that is formed by the combination of elements using fixed amounts
molecule The smallest physical unit of a substance that can exist independently
mixture When two or more substances are not joined chemically
H2O Know the formula for water
solution A homogeneous mixture of two or more substances
Ways to Separate Mixtures 1. Sifting-Using a sifter to separate two items. One item is bigger than the other.
Ways to Separate Mixtures 2. Magnetism-using a magnet to separate a magnetic object and a non-magnetic object
Ways to Separate Mixtures 3. evaporation-boiling the liquid off of a substance
Ways to Separate Mixtures 4. filtration- using a filter to separate a mixture. One object is usually larger than the other