Weak Acids and Bases
210 likes | 540 Views
Weak Acids and Bases. Today’s Class. Weak Acids Weak bases Percent dissociation. Acids (from last class).

Weak Acids and Bases
E N D
Presentation Transcript
Today’s Class • Weak Acids • Weak bases • Percent dissociation
Acids (from last class) • When an acid dissolves in water, a proton (hydrogen ion) is transferred to a water molecule to produce a hydroxonium ion and a negative ion depending on what acid you are starting from. • In the general case . . . HA + H2O H3O+ + A- • The strength of an acid is defined by the equilibrium position of the dissociation reaction shown above
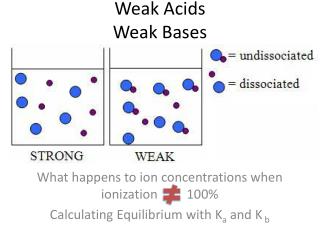
Weak Acids • A weak acid is an acid whose equilibrium lies far to the left. • Most of the acid originally present in the solution is still present as HA at equilibrium. • In other words, a weak acid only dissociates to a small extent in aqueous solution. • In contrast to a strong acid, the conjugate base of a weak acid is a much stronger base than H2O
Weak acids Example • Acetic acid is a typical weak acid. It reacts with water to produce hydronium ions and ethanoate ions, but the back reaction is more successful than the forward one. The ions react very easily to reform the acid and the water. CH3COOH + H2O CH3COO- + H3O+ • At any one time, only about 1% of the acetic acid molecules have converted into ions. The rest remain as simple acetic acid molecules • Most organic acids are weak. Hydrofluoric acid is a weak inorganic acid that you may come across elsewhere.
Strong acid • Weak acid
Weak Acids • Because of the equilibria involved, the treatment of weak acid solutions with regard to [H+] and pH is not as straightforward strong acids • Keep in mind that the solution is always in equilibrium, so we use another form of K, (equilibrium constant) called an acid dissociation constant, denoted by Ka, is an equilibrium constant for the dissociation of a weak acid
Weak Acids • Recall the expression for dissociation of an acid is: HA + H2O H3O+ + A- So the Ka expression would be Ka= [H3O+][A-] [HA]
Calculating pH of a weak acid • Let’s do an example to demonstrate Calculate the pH of 1.0M solution of HF the Ka can be found on the table on page 639 and equals 7.1x10-4 The first step is always write the major species in the solution. Since the Ka for HF is so small we know it is a weak acid and will be dissociated only to a small extent So the major species in solution are: HF and H2O
Example continued • The second step is to decide which of the major species can furnish H+ ions. In this case both species can do this: HF(aq) H+(aq) + F-(aq) Ka=7.1x10-4 H2O(l) H+(aq) + OH-(aq) Kw=1.0x10-14 • In aqueous solutions, one source of H+ ions can usually be singled out as dominant. • By comparing Kw and Ka we can see that HF, although weak, is still a much stronger acid than H2O. • We ignore the contriution of water because it is so tiny
Example Continued • From previous slide we found that the dissociation of HF will determine the equilibrium concentration of H+ and the pH: • HF(aq) H+(aq) + F-(aq) • The equilibrium expression is: • Ka=7.1x10-4=[H+][F-] [HF]
Example Continued • To solve the equilibrium problem we follow the same procedure we used in the previous chapter on equilibrium. (ICE box way) • First list the initial concentrations: [HF]0=1.0M, [F-]0=0, and [H+]0≈0 • Second step is to determine the change required to reach equilibrium • Since some of HF will dissociate to come to equilibrium (amount presently unknown) we’ll let x be the change in concentration of HF needed to reach eqilibrium. So x mol/L of HF will dissociate to produce x mol/L of H+ and x mol/L of F-. Defined in terms of x the equilibrium concentrations are: [HF]= [HF]0-x= 1.00-x [F-]=[F-]o+x= 0+x= x [H+]=[H+]o+x= 0+x= x
Example Continued • Now we can substitute equilibrium concentrations into equilibrium expression, which gives: Ka=7.1x10-4=[H+][F-]= (x)(x) [HF] 1.00-x This produces a quadratric equation that can be solved, but since the Ka for HF is so small, HF will only dissociate slightly, thus x is very small compared to 1.00 so the term in the denominator can be approximate as: 1.00-x≈1.00 So the equilibrium expression becomes: 7.2x10-4=(x)(x) 1.00 Which gives x2=7.1x10-4(1.00), taking the square root of the right side we obtain: x=2.7x10-2
Example Continued • How valid is the approimation [HF]=1.00?? • We use the following test: Compare the sizes of x and [HF]0 If the expression: x X100% is less than [HF]0 Or equal to 5%, the value of x is small enough for the approximation.
Example Continued • So testing it for our example we havex X100%=2.7x10-2 X100%=2.7% [HF]0 1.00 Therefore the approximation made was valid so the value of x can be used. Finally: x=[H+]=2.7x10-2M and pH=-log(2.7x10-2)=1.57
Summary: Solving weak acid equilibrium problems • List major species in the solution • Choose species that can produce H+, and write balanced equations for them • Compare the equilibrium constant for the reactions you have written, decide which reaction will dominate in production of H+ • Write equilibrium expression for dominant reaction • Define the change needed to reach equilibrium (define x) • Write equilibrium concentration in terms of x • Substitute the equilibrium concentrations into expression • Solve for x the easy way(assume [HA]0-x≈[HA]0) • Verify the approximation(5% rule) • Calculate [H+] and pH
Weak Bases • Much the same as weak acids: • A weak base is a base whose equilibrium lies far to the left. • Most of the base originally present in the solution is still present as original base at equilibrium. • In other words, a weak base only dissociates to a small extent in aqueous solution. • In contrast to a strong base, the conjugate acid of a weak base is a much stronger acid than H2O
Weak Bases • Weak base calculations are done in the exact same way as weak acids • The only difference is instead of using Ka as the equilibrium constant we use Kb
Percent Dissociation • It is often useful to specify the amount of weak acid that has dissociated in reaching equilibrium • It is defined as % dissocation= amount dissociated(mol/L) X100% initial concentration(mol/L) • For a given weak acid, the % dissociation Increases as the acid becomes more dilute • The % dissociation Increases as [HA]0 decreases
Example • Taking the values obtained from the previous example for weak acid % dissociation=2.7x10-2M x 100% 1.00M = 2.7%