Electrochemistry
200 likes | 314 Views
This comprehensive overview of electrochemistry explores the interdependence of chemical and electrical energy. It delves into electrochemical reactions, focusing on oxidation and reduction processes where electrons are transferred. Different types of cells, such as galvanic and electrolytic, are examined, highlighting their components and functions. The concept of cell potential and standard reduction potentials is explained, emphasizing their significance in spontaneous and non-spontaneous reactions. Additionally, key equations, including the Nernst equation, are introduced to calculate potentials under varying conditions.

Electrochemistry
E N D
Presentation Transcript
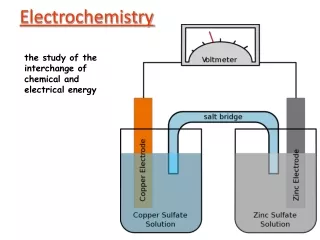
Electrochemistry The study of the interchange of chemical and electrical energy.
Electrochemical Reactions • All electrochemical reactions involve the transfer of electrons and are therefore, oxidation-reduction reaction. • Electrons are transferred from the reducing agent to the oxidizing agent. • Oxidation is a loss of electrons (increase in oxidation number)-”OIL” • Reduction is a gain of electrons (decrease in oxidation number)-”RIG”
Types of Electrochemical Cells • Galvanic or Voltaic Cells-those in which a spontaneous chemical reaction produces an electrical current that can be used to do work. • Electrolytic Cells-those in which electrical energy from an outside source causes a nonspontaneous reaction to occur.
Components of a Galvanic Cell • Cell-the reacting system • Electrodes-surfaces where the electric current exits or enters 1) anode- electrode compartment in which oxidation occurs. “AN OX” 2) cathode-electrode compartment in which reduction occurs. “RED CAT” • Salt Bridge- U-tube filled with an electrolyte or a porous disk in a tube connecting the two solutions. • Wire-path by which the electrons flow from one compartment to the other. • Electrons flow through the wire from the reducing agent to the oxidizing agent (from the anode to the cathode)
Cell Potential • Cell potential (Ecell) or electromotive force (emf) is the “pull” or driving force on the electrons. • The unit of electrical potential is the volt (V) which is defined as 1 joule/coulomb. • Cell potential is measured with a voltmeter.
Standard Reduction Potentials • Reactions in galvanic cells are broken down into half-reactions with each being assigned a reduction potential. • All half reactions are assigned reduction potentials using the standard hydrogen electrode as the reference. (see page 796) • The potentials are all given as reduction processes. • If the process must be reversed (oxidation process), the sign for the potential is reversed. • Since reduction potential is an intensive process (doesn’t depend on the how many times the reaction occurs), the value of the reduction potential is not changed when a half-reaction is multiplied by an integer to balance an equation.
Standard Reduction Potentials (continued) • The more positive the Eo value for a half-reaction, the greater tendency for the half-reaction to occur. • The more negative the Eo value for a half-reaction, the greater tendency for the half-reaction to occur in the opposite direction. • If Eocell> 0 (positive), the forward reaction is spontaneous. • If Eocell < 0 (negative), the forward reaction is not spontaneous and would have to be carried out in an electrolytic cell.
Complete the practice problems on page 797. • A. 0.71 V • B. 0.32 V
The Zinc-Copper Cell • Example: Zn(s) + Cu2+(aq) Zn2+ (aq)+ Cu(s) • Anode: Zn Zn2+ + 2 e- • Cathode: Cu2+ + 2e- Cu • Eocell = .337 + .763 = 1.10 V • Line Notation: Zn | Zn2+ | | Cu2+ | Cu (anode is written on the left side and the vertical line represents a phase difference or boundary)
Write the line notation for a galvanic cell consisting of copper (II) and chromium (III) • Cr3+ + 3e- Cr Eo = -0.74 V • Cu2+ + 2e- Cu Eo = 0.337 V • Copper reduction occurs at the cathode • Chromium oxidation occurs at the anode. • Line Notation: Cr | Cr3+ | | Cu2+ | Cu
Cell Potential, Work, and Free Energy • The work that can be accomplished when electrons are transferred through a wire depends on the “push” behind the electrons. • Potential difference (V) = work (J)/charge (C) • E = -w/q • Work is viewed from the point of view of the system. (Work flowing out of a system is indicated by a minus sign). • In any real, spontaneous process some energy is wasted due to frictional heating-the actual work realized is always less than the calculated maximum.
Electrical Charge • The charge on one mole of electrons is a constant called the faraday (F), which has the value 96,485 coulombs of charge per mole of electrons. • q = nF (n is the number of moles of e-) • w (∆G) = -nFEmax • Solve example 17-3 on page 802.
The Nernst Equation • The Nernst Equation is used to calculate electrode potential and cell potentials for concentrations and partial pressures other than standard-state values. • E = Eo – (2.303 RT/nF ) log Q • E = potential under nonstandard conditions • Eo = standard potential • R= 8.314 • T = temp in Kelvin • n= number of moles of electrons transferred • F = 96,485 C/mole of e- • Q=reaction quotient
Electrolytic Cells • In an electrolytic cell, an outside source of voltage is used to force a nonspontaneousredox reaction to take place. • Oxidation takes place at the anode and reduction takes place at the cathode just as it does in a galvanic cell. • The cell potential in an electrolytic cell < 0. • Electrolytic cells are used in electroplating.
Electrolysis Problems • I = q/t • I = current (amperes, A) • 1 amp = 1C/sec • q = charge (coulombs, C) • t = time (sec) • Once the charge is known, solve the problem as a stoichiometry problem.
Practice Problem #1 • How long must a current of 5.00 A be applied to a solution of Ag+ toproduce 10.5 g of silver metal?
Practice Problem #2 • What mass of Co can be produced from aqueous Co2+ in 1 hour with a current of 15 A?
Practice Problem #3 • A zinc-copper battery is constructed as follows at 25oC: • Zn Zn2+ (0.1M) Cu2+ (2.50M) Cu The mass of each electrode is 200g. • Calculate the cell potential when this battery is first connected. • Calculate the cell potential after 10.0A of current has flowed for 10.0 h. • Calculate the mass of each electrode after 10.0 h • How long can this battery deliver a current of 10.0A before it goes dead?