Chapter 17: Electrochemistry
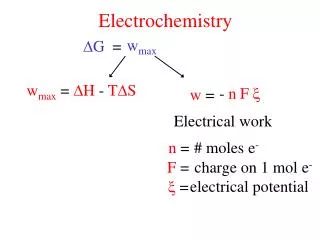
Chapter 17: Electrochemistry. Review of Redox Reactions Galvanic Cells: Using spontaneous redox reactions to generate electrical energy. Galvanic Cells Cell potential D G and work Cell potential and concentration Applications: Batteries, fuel cells, corrosion

Chapter 17: Electrochemistry
E N D
Presentation Transcript
Chapter 17: Electrochemistry • Review of Redox Reactions • Galvanic Cells: Using spontaneous redox reactions to generate electrical energy. • Galvanic Cells • Cell potential • DG and work • Cell potential and concentration • Applications: Batteries, fuel cells, corrosion • Electrolytic Cells: Using electricity to cause nonspontaneous redox reactions to occur. • Electrolytic Cells • Applications: Electrolysis of water, electrolysis of mixtures of ions, production of Al, electrorefining, metal plating, electrolysis of NaCl
Example 1 Balance the following redox reactions using the half reaction method. • ClO3- + As2S3 Cl- + H2AsO4- + SO42- • K2S + KMnO4 S8 + MnO2 + KOH
Example 2 If a standard Cu|Cu2+ electrode is connected to a standard Al|Al3+ electrode, what reaction occurs? What is °cell? Sketch the cell, label the cathode and anode, show the direction of electron flow, and give the line notation for the cell.
Example 3 Consider the following species under standard conditions: Ce4+, Ce3+, Fe2+, Fe3+, Fe, Mg, Mg2+, Ni2+, Sn • Which is the strongest oxidizing agent? • Which is the strongest reducing agent? • Will Fe dissolve in 1.0 M Ce4+? If so, will Fe3+ or Fe2+ be formed? • Which can be oxidized by H+(aq)? • Which can be reduced by H2(g)?
Example 4 Select an oxidizing agent to oxidize Cl- to Cl2 without oxidizing Br- to Br2.
Example 5 Calculate the maximum work available from 25.0 g of aluminum in the following galvanic cell for which the emf is 1.15 V. Note that O2 is reduced to H2O in this reaction. Al(s)|Al3+(aq)||H+(aq)|O2(g)|Pt(s)
Example 6 Calculate the cell potential for the following Galvanic cell at 25°C. Ni(s)|Ni2+(1.0M)||Sn2+(1.0x10-4M)|Sn(s)
Example 7 Find the potential of a Ag+(1.0x10-7M)|Ag(s) electrode at 25°C.
Example 8 Calculate the equilibrium constant for the following reaction at 25°C. Ag+(aq) + Fe2+(aq) Ag(s) + Fe3+(aq)
Example 9 A concentration cell is made up of two Ag/Ag+ half cells. In the first half cell, [Ag+] = 0.010 M. In the second half cell, [Ag+] = 4.0 x 10-4 M. What is the cell potential? Which half cell functions as the anode?
Example 10 Predict the products and calculate the minimum voltage required for the electrolysis of the following substances using platinum electrodes. • MgBr2(l) • 1.0 M NiCl2(aq)
Example 11 Write the net ionic equation for the reaction you expect to occur when the electrolysis of NiSO4(aq) is conducted using a nickel anode and an iron cathode.
Example 12 How many grams of silver are deposited at a platinum cathode in the electrolysis of an aqueous solution of AgNO3 by 1.73A of electric current in 2.5 hours?
Example 13 How long will it take to produce 10.0 g of bismuth (Bi) by the electrolysis of a BiO+ solution using a current of 25.0 A?
Figure 17.25: Downs cell for production of sodium and chlorine