PHOSPHORUS CYCLE
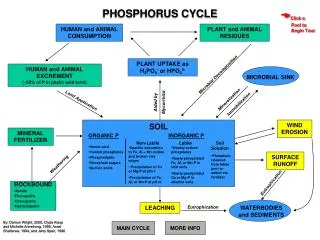
Land Application. PHOSPHORUS CYCLE. Click a Pool to Begin Tour. HUMAN and ANIMAL CONSUMPTION. PLANT and ANIMAL RESIDUES. PLANT UPTAKE as H 2 PO 4 - or HPO 4 2-. HUMAN and ANIMAL EXCREMENT (~50% of P in phytic acid form). Microbial Decomposition. MICROBIAL SINK.

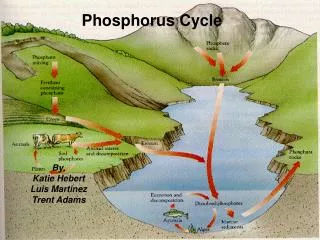
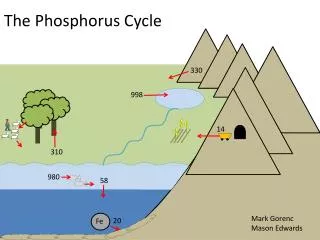
PHOSPHORUS CYCLE
E N D
Presentation Transcript
Land Application PHOSPHORUS CYCLE Click a Pool to Begin Tour HUMAN and ANIMAL CONSUMPTION PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- HUMAN and ANIMAL EXCREMENT(~50% of P in phytic acid form) Microbial Decomposition MICROBIAL SINK Mineralization Immobilization Aided by Mycorrhiza SOIL WIND EROSION MINERALFERTILIZER ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer SURFACE RUNOFF Weathering Eutrophication • ROCKBOUND • Apatite • Fluorapatite • Chlorapatite • Hydroxlapatite LEACHING Eutrophication WATERBODIES and SEDIMENTS By: Damon Wright, 2000; Clyde Alsup and Michelle Armstrong, 1998; Asrat Shaiferaw, 1994; and Jerry Speir, 1996 MAIN CYCLE MORE INFO
Land Application PHOSPHORUS CYCLE PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- HUMAN and ANIMAL EXCREMENT(~50% of P in phytic acid form) Microbial Decomposition MICROBIAL SINK Mineralization Immobilization Aided by Mycorrhiza SOIL WIND EROSION MINERALFERTILIZER ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer SURFACE RUNOFF Weathering • ROCKBOUND • Apatite • Fluorapatite • Chlorapatite • Hydroxlapatite LEACHING MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE SOIL MINERALFERTILIZER ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer Weathering • ROCKBOUND • Apatite • Fluorapatite • Chlorapatite • Hydroxlapatite MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE SOIL MINERALFERTILIZER ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer • ROCKBOUND • Apatite • Fluorapatite • Chlorapatite • Hydroxlapatite MAIN CYCLE MORE INFO
Land Application PHOSPHORUS CYCLE HUMAN and ANIMAL CONSUMPTION HUMAN and ANIMAL EXCREMENT(~50% of P in phytic acid form) SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE HUMAN and ANIMAL CONSUMPTION PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- HUMAN and ANIMAL EXCREMENT(~50% of P in phytic acid form) MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE HUMAN and ANIMAL CONSUMPTION PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- Aided by Mycorrhiza SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE HUMAN and ANIMAL CONSUMPTION PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- Microbial Decomposition SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE MICROBIAL SINK Mineralization Immobilization SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE SOIL WIND EROSION ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer WATERBODIES and SEDIMENTS MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer SURFACE RUNOFF Eutrophication WATERBODIES and SEDIMENTS MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE WIND EROSION SURFACE RUNOFF Eutrophication LEACHING Eutrophication WATERBODIES and SEDIMENTS MAIN CYCLE MORE INFO
PHOSPHORUS CYCLE SOIL ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer LEACHING Eutrophication WATERBODIES and SEDIMENTS MAIN CYCLE MORE INFO
Aided by Mycorrhiza Weathering Eutrophication Eutrophication PHOSPHORUS CYCLE Click a Pool to SearchWeb Links HUMAN and ANIMAL CONSUMPTION PLANT and ANIMAL RESIDUES PLANT UPTAKE as H2PO4- or HPO42- HUMAN and ANIMAL EXCREMENT(~50% of P in phytic acid form) Microbial Decomposition MICROBIAL SINK Mineralization Land Application Immobilization SOIL WIND EROSION MINERALFERTILIZER ORGANIC P INORGANIC P Non-Labile Labile Soil Solution • Humic acid • Inositol phosphates • Phospholipids • Phosphate sugars • Nucleic acids • Specific adsorption to Fe, Al,+ Mn oxides and broken clay edges • Precipitation of Ca or Mg-P at pH>7 • Precipitation of Fe, Al, or Mn-P at pH<5 • Weakly sorbed phosphates • Newly-precipitated Fe, Al, or Mn-P in acid soils • Newly precipitated Ca or Mg-P in alkaline soils • Phosphate released from labile pool or added via fertilizer SURFACE RUNOFF • ROCKBOUND • Apatite • Fluorapatite • Chlorapatite • Hydroxlapatite LEACHING WATERBODIES and SEDIMENTS By: Damon Wright, 2000; Clyde Alsup and Michelle Armstrong, 1998; Asrat Shaiferaw, 1994; and Jerry Speir, 1996 START AGAIN MORE INFO
Form taken up by plant: H2PO4-, HPO4= Mobility in soil: None; roots must come in direct contact with orthophosphate P Mobility in plant: Yes Deficiency symptoms: Lower leaves with purple leaf margins Deficiency pH range: <5.5 and >7.0 Toxicity symptoms: None Toxicity pH range:Non toxic (optimum availability pH 6.0-6.5) Role in plant growth: Important component of phospholipids and nucleic acids (DNA and RNA) Role in microbial growth: Accumulation and release of energy during cellular metabolism Concentration in plants: 1,000 – 5,000 ppm (0.1 –0.5%) Effect of pH on availability: H2PO4- at pH < 7.2 HPO4= at pH > 7.2 Interactions with otherP x N, P x Zn at high pH, in anion nutrients: exchange P displaces S, K by mass action displaces Al inducing P deficiency (pH<6.0) BACK TO MAIN CYCLE Additional Information on Phosphorus MORE INFO
BACK TO MAIN CYCLE Additional Information on Phosphorus (Cont.) • P fertilizer sources: Rock phosphate, phosphoric acid, Ca orthophosphates, ammoniumphosphates, ammonium poly-phosphates, nitric phosphates, K phosphates, microbial fertilizers (phosphobacterins) increase P uptake • Additional categories: • Mineralization/ C:P ratio of < 200: net mineralization of immobilization: organic P; C:P ratio of 200-300: no gain/loss of inorganic P; C:P ratio of >300: net immobilization of inorganic P • P fixation: Formation of insoluble Ca, Al, and Fe phosphates • Al(OH)3 + H2PO4- Al(OH)2HPO4 • (Soluble) (Insoluble) • Organic P sources: Inositol phosphate (Esters of orthophosphoric acid), phospholipids,nucleic acids, phosphate sugars • Inorganic P sources: Apatite and Ca phosphate (unweathered soils) and Fe and Al sinks from P fixation (weathered soils) • Waste: Poultry litter (3.0 to 5.0%), steel slag (3.5%), electric coal ash (<1.0%) MORE INFO
BACK TO MAIN CYCLE Additional Information on Phosphorus (Cont.) • Total P levels in soil: 50 – 1500 mg/kg • Solution concentration range: < 0.01 to 1.0 ppm • Applied fertilizer: < 30% recovered in plants, more P mustbe added than removed by crops References: • Alexander, M., 1977. Introduction to Soil Microbiology. 2nd Edition. John Wiley and Sons, NY. • Brady, N.C., 1990. The Nature and Properties of Soils. 10th Edition. Macmillan Publishing Co., NY. • Brigham Young University. 1997. The Phosphorus Cycle. http://ucs.byu.edu/bioag/aghort/214pres/geochem.htm • Harrison, A.F., 1987. Soil Organic Phosphorus. A Review of World Literature. C.A.B. p.39. • Pierre, W.H., 1948. The Phosphorus Cycle and Soil Fertility. J. Amer. Soc. of Agron., 40:1-14. • Pierzynski, G.M., Sims, J.T., and Vance, G.F., 1994. Soil and Environmental Quality. Lewis Publishers, FL. • Stewart, J.W.B., and Sharpley, A.N., 1987. Controls on Dynamics of Soil and Fertilizer Phosphorus and Sulfur in Soil Fertility and Organic Matter as Critical Components of Production Systems, SSSA Spec. Pub. No.19, 101-121. • Tiessen, H., 1995. Phosphorus in the Global Environment – Transfers, Cycles and Management. John Wiley and Sons, NY. • Tisdale, S.L., Nelson, W.L., Beaton, J.D. and Havlin, J.L., 1993. Soil Fertility and Fertilizers. Macmillan Publishing Co., NY.