Water
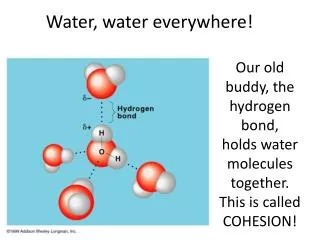
Water H 2 O, Sea Water and what it means to life in the ocean…. Figure 3.01 Hydrogen Bonds, Between water molecules Are weak, but IMPORTANT! States of water Water occurs as solid, liquid & gas naturally No hydrogen bonds in water vapor

Water
E N D
Presentation Transcript
Water • H2O, Sea Water and what it means to life in the ocean….
Figure 3.01 Hydrogen Bonds, Between water molecules Are weak, but IMPORTANT!
States of water • Water occurs as solid, liquid & gas naturally • No hydrogen bonds in water vapor • Some molecules joined by hydrogen bonds in liquid – bonds break & reform • All molecules rigidly bonded in solid
Heat and Water • Adding heat makes molecules vibrate • Heat energy is used to cause vibration AND to break hydrogen bonds
Heat and Water • Ice melts at relatively high temperatures compared to similar substances • (OoC instead of –90oC)
Water = Universal Solvent • Good at dissolving salts (the solutes) • Salts exist as separate ions in seawater
Seawater • Salts come from weathering of rock or outgassing from hydrothermal vents • Sodium and Chloride account for 85% of the dissolved solids (solutes) in sea water. • This makes it various degrees of SALINE: “total amount of salt dissolved in sea water”
SeaWater • WHERE does the “salt” come from? • And WHAT else is in the water?
Salinity • The total amount of dissolved salts in seawater • Measured in parts per thousand (ppt or o/oo) • Average salinity of sea water is 35%
Density of (sea)water • Density also affects the salinity of water • Liquid water gets denser as the temperature decreases • BUT ice is LESS dense than liquid water • Increases with decreasing temperature • Increases with increasing salinity
Temp. of ocean surface: cold = blue, warm = red Typical Ocean temp. range are 28-86 degrees F
Stratified Ocean (3 Layers) • Salinity, Temperature and Density all effect ocean waters giving OCEAN GRADIENTS or stratified layers. • Dense, cold water @ bottom • Less dense, warmer water @ surface (floating on layer below)
3 layered Ocean • Surface “mixed” layer: 100-200 m thick • Mid “intermediate” layer: 200-1500 m thick contains the main thermocline (transition zone between warm and cold top/bottom layers) • Deep “bottom” layer: after 1500 m
Light in the ocean • Transparency depends on what is suspended in the water • Different colors penetrate to different depths
Pressure • Water is heavier than air • Pressure changes ( a lot!) w/ increased water depth • 1 atm. of pressure = sea level (on land) but in ocean each 10 m of depth (33’) you add another atm. of pressure
Water, salinity, light, pressure, depth • How does this affect marine “life?”
Life in the Ocean • Deal with challenges that are unique to marine environment • Must maintain suitable conditions inside the organism’s body
Figure 4.13a More salt in cell, Water diffuses in to cell, it will swell and burst.
Figure 4.13c More salt outside cell, Water moves out of cell, it will shrivel.
Hawksbills’ “excrete” glands (near eyes) to get rid of excess salt
Figure 3.14 Groupers swim bladder has expanded, due to pressure, thus stomach has Been forced through mouth!
Buoyancy Two deep-sea fishes on the deck of a ship after being hauled up from a depth of 800 m. Both fishes were seriously damaged and distorted by the rapid expansion of gases in their swim bladders as they were brought to the surface.
Buoyancy Fnft: A physoclistous swim bladder and associated blood vessels.
Temperature • How does this effect marine organisms? • It dictates where they live and metabolize
Temperature Regulation • Most marine animals are ectotherms (“cold-blooded”)
Temperature Regulation • Most marine animals are ectotherms (“cold-blooded”) • Mammals and birds are endotherms • Use fat, feathers for insulation
Temperature • Most marine animals are adapted to living at a specific temperature • Temperature determines species ranges
New Topic • Tides…
Tides • Tides • Tide Tables • Waves • Weather (resultant)