Chemical Equilibrium
320 likes | 528 Views
Chemical Equilibrium. Chapter 13. 13.1 The Equilibrium Condition .

Chemical Equilibrium
E N D
Presentation Transcript
Chemical Equilibrium Chapter 13
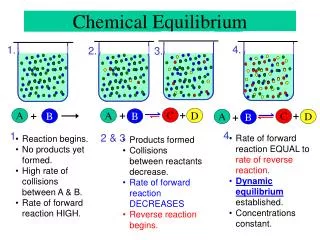
13.1 The Equilibrium Condition • So far, we have assumed that reactions proceed to completion. Many actually fall short of completion and reach chemical equilibrium. Chemical equilibrium is the state where the concentrations of all reactants and products remain constant with time. Figure 13.1 a-d A Molecular Representation of the Reaction 2NO2(g) - N2O4(g)
If the reaction appears to have gone to completion, the equilibrium position will favor the products and lies to the right (in the direction of the products). • If the reaction only occurs to a small extent, where the reaction is almost undetectable, then the equilibrium position favors the reactants and lies far to the left (in the direction of the reactants). H2O (g) + CO (g) ↔ H2 (g) + CO2(g)
The concentration of the product increases and the concentration of the reactant decreases, but at a certain time, the concentrations stay the same. Notice how the [reactant] never reaches 0. N2(g) + 3H2(g) ↔ 2NH3 (g)
Remember that reactions occur when molecules collide into one other with enough energy and in the correct orientation. Higher concentrations = more collisions= faster reactions.
Factors determining the equilibrium position: • Initial concentrations • Energies of reactants and products • “Organization” of reactants and products • Concentrations of reactants and products may remain unchanged when mixed if: • The system has reached equilibrium • Forward and reverse reactions are so slow that the system moves to equilibrium at an undetectable rate.
13.2 The Equilibrium Constant • Law of mass action: jA + kB ↔ lC + mD • Where j, k, l, m are coefficients and A, B, C, D are the chemical species. • The law of mass action is represented by the equilibrium expression: • K is the equilibrium constant and does not have units. Example 13.17 on pg. 614
The value of the equilibrium constant can be calculated if equilibrium concentrations are known for a given reaction. • Example 13.21 and 13.23 on pg. 615
The equilibrium expression for a reaction is the reciprocal of that for the reaction in reverse. • When a balanced equation is multiplied by a factor n, the equilibrium expression for the new reaction is the old reaction raised to the nth power. njA + nkB ↔ nlC + nmD • Example 13.19 on pg. 614
Although the equilibrium expression is constant for a given reaction, equilibrium concentrations will NOT always be the same. • Each set of equilibrium concentrations is called an equilibrium position. • Equilibrium positions can vary, but equilibrium constants do not vary.
13.3 Equilibrium Expressions Involving Pressures • PV=nRT, therefore P=(n/V)RT=CRT • Since, C is directly related to P
K and Kc are used for the equilibrium constant in terms of concentration. Kp represents the equilibrium constant in terms of partial pressures. • Where n=(l+m)-(j+k) which is the difference in the sums of the coefficients • Example 13.25, 13.27 on pg. 615
13.4 Heterogeneous Equilibria • Homogeneous equilibria is for systems where reactants and products are in the same gaseous phase. • Heterogeneous equilibria involves reactions involving more than one phase. CaCO3 (s)↔ CaO(s) + CO2(g)
Positions of heterogeneous equilibrium does not depend on the amounts of pure solid or liquid present. Therefore, their concentrations are NOT included in the equilibrium expression. 2H2O (l) ↔ 2H2 (g) +O2 (g) Kc=[H2]2[O2] and Kp=(PH22)(Po2) • Examples 13.29 and 13.31 pg. 615
13.5 Applications of the Equilibrium Constant • Equilibrium Constants helps predict: • The tendency of the reaction to occur (not rate). • Whether a given set of concentrations represent an equilibrium condition. • The equilibrium position achieved from a certain set of initial concentrations.
For this reaction: Initial Concentrations Equilibrium Concentrations
The Extent of Reaction • Is indicated by the value of K • If K is much greater than 1, mostly products are present and the equilibrium lies to the right. • If K is very small, the reaction does not occur to a significant extent and the equilibrium is far to the left. • Remember that the size of K and rate are NOT directly related.
Reaction Quotient • If the concentrations of a reactant or product is zero, the system will shift to produce the missing component. • To determine this shift, we use the reaction quotient, Q. • It is the same as the equilibrium constant, except initial concentrations are used rather than equilibrium concentrations.
To determine the direction of shift to reach equilibrium, compare K and Q. • If Q=K, the system is at equilibrium and no shift will occur. • If Q>K, the ratio of initial products to initial reactants is too large. The system will shift left to form more reactants and achieve equilibrium. • If Q<K, the ratio of initial reactants to initial products is too large. The system will shift right to form more products and achieve equilibrium. Example 13.33 on pg. 615
Calculating Equilibrium Pressures and Concentrations • Initial Concentration • Change in Concentration • Equilibrium Concentration ICE
13.6 Solving Equilibrium Problems • Write the balanced reaction. • Write the equilibrium expression. • List the Initial concentrations. • Calculate Q, and determine the direction of the shift of equilibrium (do not have to do if you have 0 concentration of a substance). • Define the change needed to reach equilibrium, and apply this change to the initial concentrations to determine the equilibrium concentrations. • Substitute the equilibrium concentration into the equilibrium expression, and solve for the unknown. • Check your calculated equilibrium concentration by making sure they give the correct value for K.
If K is really small (10-5), x will represent a really small number and can be disregarded in comparison to the reactant. Examples 13.33 and 13.37-13.51odd on pg. 615-617
13.7 Le Chậtelier’s Principle • Manufacturers want to ensure that equilibrium lies far to the right, in favor of the production of products. • We can predict the effects of changes in concentration, pressure, and temperature based on Le Chậtelier’s Principle, which says that if a change is imposed on a system at equilibrium then the position of the equilibrium will shift in order to reduce that change.
The Effect of Change on Concentration • If add a reactant, the equilibrium position will shift to the right in order to reduce the amount of reactant. • If a product is added, the equilibrium position will shift to the left in order to reduce the amount of product. • If a substance is removed, the equilibrium position will shift towards that substance in order to increase the amount of the removed substance.
As4O6(s) + 6C(s) → As4(g) + 6CO(g) Predict the direction of the shift of the equilibrium position in response to each of the following changes in conditions. a.) Addition of carbon monoxide b.) Addition of tetraarsenic hexoxide c.) Removal of carbon d.) Removal of gaseous arsenic
The Effect of a Change in Pressure Three ways to change the pressure: • Add or remove a gaseous reactant or product. -already discussed • Add an inert gas. -do not participate in the reaction in any way; therefore, they do not affect the equilibrium • Change the volume of the container. -discussed on the next slide
Since volume is directly proportional to moles according to PV=nRT, the system will reduce the total number of molecules present by shifting towards the side of the reaction with the least number of moles when the volume is decreased.
Predict the shift in equilibrium position that will occur for each of the following processes when the volume is reduced. a.) P4(s) + 6Cl2(g) → 4PCl3(l) b.) PCl3 (g) + Cl2 (g) → PCl5 (g) c.) PCl3 (g) + 3NH3 (g) → P(NH2)3 (g) + 3HCl (g)
Changes in pressure and concentrations do NOT change K, but temperature changes alter the value of K. Endothermic reactions can be viewed as adding an additional reactant. The equilibrium shifts to the right, increasing K. Exothermic reactions can be viewed as adding a product. The equilibrium shifts to the left, decreasing K. The Effect of a Change in Temperature
For the reaction: 58 kJ +N2O4(g) →2NO2(g) Examples 13.57-13.63 on pg. 617-618 Summary of Le Chậtelier’s Principle