Solution Concentration
This guide explores the concept of solution concentration, specifically focusing on how to express it in terms of percent mass. Concentration is defined as the quantity of solute dissolved in a specific amount of solvent, calculated using the formula: Concentration = (mass of solute / mass of solution) × 100. Practice problems include calculating the percent concentration of sucrose in solutions with varying amounts of solute and solvent, enhancing your understanding of concentration in practical scenarios.

Solution Concentration
E N D
Presentation Transcript
Solution Concentration Percents, pph, ppm
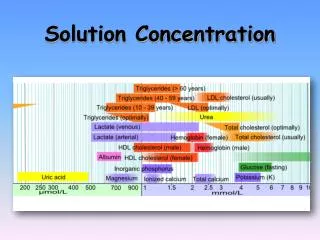
Solution Concentration • Quantity of solute dissolved in a specific amount of solvent • Concentration = mass of solute ------------------------------ X 100 Mass of solution Hint: solution = solute + solvent
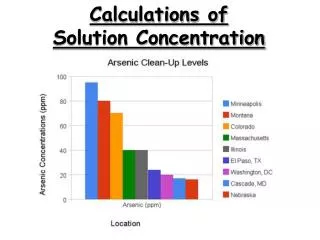
Practice Calculations • What is the concentration of the following solution expressed as percent sucrose by mass? 25 g of sucrose is dissolved in 175 g of water.