Enzyme Kinetics
Enzyme Kinetics. The aims of enzyme kinetics studies are to: Measure the rates of enzyme catalyzed reactions Examine enzyme specificity Identify selective and potent enzyme inhibitors Determine the kinetic mechanism of an enzyme. Enzyme Activity Measurements.

Enzyme Kinetics
E N D
Presentation Transcript
Enzyme Kinetics • The aims of enzyme kinetics studies are to: • Measure the rates of enzyme catalyzed reactions • Examine enzyme specificity • Identify selective and potent enzyme inhibitors • Determine the kinetic mechanism of an enzyme
Enzyme Activity Measurements The rate of an enzyme-catalyzed reaction is measured by an activity assay where the formation of product (or the disappearance of substrate) is determined over a given time period The formation of product can be measured continuously or by sampling the reaction mixture at different time intervals Typical means of determining product (or substrate) concentrations are absorption or fluorescence spectroscopy for continuous assays or HPLC, GC or radiolabeled sampling for fixed time assays If neither the substrate nor the product of an enzyme have spectral signals then a coupled assay can be designed where the product is converted to something with a signal by using another enzyme
lactate NAD lactate dehydrogenase substrate ATP pyruvate NADH pyruvate kinase kinases phospho- substrate phosphoenol pyruvate ADP Coupled Enzyme Activity Assay [A340 = 6.2 mM-1 ] In this case the formation of a product (ADP) with an identical spectral signal as the substrate (ATP) can be measured by following the disappearance of a substrate for a coupling enzyme (NADH) with a unique spectral signal
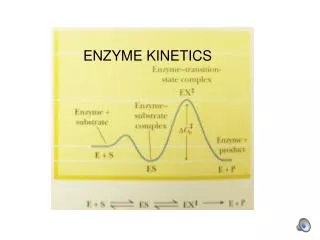
Enzyme Kinetics The velocity of an enzyme-catalyzed reaction is described by the Michaelis-Menten equation Plotting v vs. [S] yields a hyperbola Vmax is the maximum velocity This is the rate of the reaction at saturating substrate concentration Km is the Michaelis constant This is the concentration of substrate [S] that gives half of Vmax
Enzyme Kineticslimiting cases first order region
Enzyme Kineticslimiting cases zero order region
Enzyme Kinetics It is not easy to extrapolate a hyperbola to its limiting value to determine the maximum velocity The Michaelis-Menten equation can be recast into a linear form The y-intercept give the Vmaxvalue and the slope gives Vmax/Km
Enzyme Kineticstwo substrate enzymes The Michaelis-Menten equation for an enzyme with two substrates has a similar form to the one substrate equation The major difference is the presence of some additional terms What does Km measure ?
Enzyme KineticsWhat does Km measure ? If catalysis (k3) is rate limiting, only then does Km Ki and the Km value is a reasonable measure of substrate affinity
Enzyme Kineticstwo substrate enzymes If an enzyme has two substrates then they can bind to the active site in an obligatory order or they can bind in a random order An ordered kinetic mechanism Horizontal line represents the enzyme (E) A and B are the substrates P and Q are the products The brackets ( ) represent the conversion of substrates to products
Enzyme Kineticstwo substrate enzymes A fully random kinetic mechanism Reactions can also be partially ordered 1. Ordered addition of substrates, random release of products 2. Random addition of substrates, ordered release of products
Enzyme Kineticsdetermining kinetic parameters This is a plot of 1/velocity vs. 1/[substrate A] at different levels of substrate B We can’t simply extract the kinetic constants from this plot In order to get these values we have to replot the data
Enzyme Kineticsdetermining kinetic parameters From a replot of the intercepts we can determine Vmax and Kb From a replot of the slopes we can determine Ka and Kia We cannot distinguish between an ordered and a random kinetic mechanism from this initial velocity data analysis There is another type of kinetic mechanism in which the catalytic reaction can start without both substrates being present
Enzyme Kineticsping-pong mechanism In this example the first substrate (A) is converted to product (P) and the product departs before the second substrate (B) binds This leaves the enzyme in a modified form (F) which can now bind (B) and convert it to product (Q)
Enzyme Kineticsping-pong mechanism This plot gives parallel lines that are diagnostic of this mechanism Replots are again needed to determine the kinetic parameters
Enzyme Kineticsping-pong mechanism Again, the intercept replot gives us Vmax and Kb However, since all the lines have the same slope this replot only gives us Ka Kia is zero for a ping-pong mechanism
Enzyme Kineticsinhibition studies Inhibition studies will help to identify: The detailed kinetic mechanism of an enzyme (ordered or random, order of substrate addition) New potent inhibitors of an enzyme that can be developed into drugs
Enzyme Kineticsinhibition studies To conduct these studies we vary the concentration of a potential inhibitor and then plot the rates in the presence of inhibitor vs. the substrate concentration The presence of the inhibitor can affect either the slope or the intercept of the linear reciprocal plots If there is an intercept effect this means that the inhibitor combines with a different form of the enzyme than does the substrate that is being varied If there is a slope effect then the inhibitor combines with the same enzyme form as the varied substrate
Enzyme Kineticscompetitive inhibition A competitive inhibitor binds to the same enzyme form(E) as the varied substrate The slope replot gives the Ki value for the inhibitor
Enzyme Kineticsuncompetitive inhibition An uncompetitive inhibitor binds to a different enzyme form (ES) from that of the varied substrate (E) The intercept replot gives the Ki value for the inhibitor
Enzyme Kineticsnoncompetitive inhibition A noncompetitive inhibitor can bind to more than one enzyme form (E and ES) The replots gives the Kis and Kiivalues for the inhibitor
Enzyme Kineticsproduct inhibition studies Product inhibition studies can be used to distinguish between the various possible kinetic mechanisms * becomes NC for any substrate-product pair that can bind simultaneously to the enzyme (dead-end complex)
Enzyme Kineticsterreactant mechanisms Some enzymes have three substrates and/or three products 1. Synthetases 2. Hydroxylases
Enzyme Kineticsterreactant mechanisms 3. Certain Dehydrogenases 4. Carboxylases
Enzyme Kineticsterreactant mechanisms The Michaelis-Menten equation for an enzyme with three substrates has a similar overall form to the one or two substrate equation For enzymes with terreactant mechanisms we must use replots of the replots to separate and determine all of the kinetic parameters There are eight different terms in the rate equation and the presence or absence of these terms are indicative of different kinetic mechanisms
Enzyme Kineticsterreactant mechanisms • Reciprocal plots are made vs. 1/C at several levels of A • This pattern is then repeated at difference B levels • The slopes and intercepts of these plots are replotted vs. 1/A • The slopes and intercepts of these replots are then replotted vs. 1/B • A zero slope or zero intercept in these secondary replots indicate the absence of that term from the rate equation
Enzyme Kinetics • The kinetic mechanism of an enzyme gives the order of addition of substrates and release of products • Mechanisms can be ordered, random, or ping-pong • Plots (and replots) are used to determine the kinetic parameters (Vmax, Km, Vmax/Km) for each substrate • Inhibition studies help to sort out complex kinetic mechanisms, identify new enzyme inhibitors, and measure their potency (Ki)