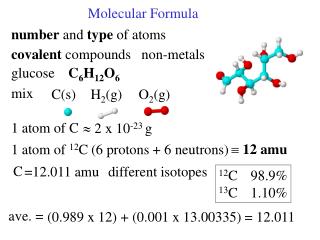
Molecular Formula
Molecular Formula. Chapter 7.7 Molecular Formula. Objective: To calculate the molecular formula for a compound. Determining a Molecular Formula from an Empirical Formula. Step 1: Find the molar mass of the empirical formula.

Molecular Formula
E N D
Presentation Transcript
Chapter 7.7Molecular Formula Objective: To calculate the molecular formula for a compound.
Determining a Molecular Formula from an Empirical Formula Step 1: Find the molar mass of the empirical formula. Step 2: Divide the molar mass of the molecular formula by the molar mass of the empirical formula. Step 3: Multiply the empirical formula by the factor obtained in step 2.
Example #1 The empirical formula for a compound is P2O5. Its experimental molar mass is 284 g/mol. Determine the molecular formula of the compound.
Example #1 The empirical formula for a compound is P2O5. Its experimental molar mass is 284 g/mol. Determine the molecular formula of the compound. Step 1: The molar mass of P2O5 is 141.94 g/mol Step 2: (284 g/mol) / (141.94 g/mol) = 2.00 Step 3: 2 x (P2O5) = P4O10
Example #2 Determine the molecular formula for a compound that has a molar mass of 232.41 g/mol and an empirical formula of OCNCl.
Example #2 Determine the molecular formula for a compound that has a molar mass of 232.41 g/mol and an empirical formula of OCNCl. Step 1:The molar mass of OCNCl is 77.47 g/mol Step 2: (232.41 g/mol) / (77.47 g/mol) = 3.00 Step 3: 3 x (OCNCl) = O3C3N3Cl3
Example #3 Determine the molecular formula for a compound that has a molar mass of 32.06 g/mol and an empirical formula of NH2.
Example #3 Determine the molecular formula for a compound that has a molar mass of 32.06 g/mol and an empirical formula of NH2. Step 1:The molar mass of NH2 is 16.01 g/mol Step 2: (32.06 g/mol) / (16.01 g/mol) = 2.00 Step 3: 2 x (NH2) = N2H4
Example #4 Determine the molecular formula for a compound that has a molar mass of 120.12 g/mol and an empirical formula of CH2O.
Example #4 Determine the molecular formula for a compound that has a molar mass of 120.12 g/mol and an empirical formula of CH2O. Step 1:The molar mass of CH2O is 30.01 g/mol Step 2: (120.12 g/mol) / (30.01 g/mol) = 4.00 Step 3: 4 x (CH2O) = C4H8O4