Ammonia
Ammonia. Jennifer Cunningham Leah Dover Ms. Wallace Chemistry Hn. 3 rd 11/19/10. N 2(g) + 3H 2(g) 2NH 3(g ) Nitrogen and hydrogen combine to perform a chemical reaction that produces ammonia. .

Ammonia
E N D
Presentation Transcript
Ammonia Jennifer Cunningham Leah Dover Ms. Wallace Chemistry Hn. 3rd 11/19/10
N2(g) + 3H2(g) 2NH3(g) Nitrogen and hydrogen combine to perform a chemical reaction that produces ammonia.
Ammonia is produced by the Haber-Bosch process, also known as the Haber-Ammonia Process and the Synthetic Ammonia Process. This process was created in 1909 by German physical chemist, Fritz Haber. Later on, Carl Bosch continued to develop the process so that it would be economically possible. Their work won them both a Nobel Prize; Haber in 1918 for its creation, and Bosch in 1931 for developing high-pressure conditions that obtained a higher yield economically.
Today, China is the number one producer of ammonia; followed by India, Russia, and the U.S. There are several buyers all over the world, because it is used in many everyday products. Mainly, ammonia is used in fertilizers, which is where 80% of its production goes. It is also used as a precursor for food and it even can be linked as a building block for the synthesis of several pharmaceuticals. In addition, ammonia is used commercially in cleaners and has minor/emerging uses in things such as refrigeration, fuel, stimulants, textiles, and woodworking.
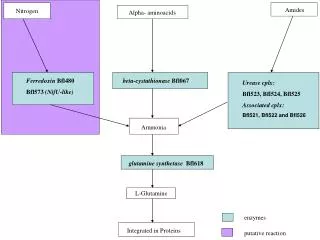
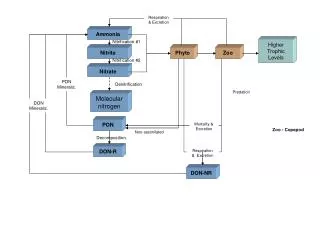
The reactants for producing ammonia are nitrogen and hydrogen. Nitrogen makes up about 80% of the Earth’s atmosphere and continuously goes through the nitrogen cycle. This cycle is crucial to life and involves nitrogen being constantly decomposed and reproduced by living organisms. Hydrogen is the most abundant element in the world and makes up about the other 20% of the Earth’s atmosphere. It can be produced from several different things such as natural gas, coal, biomass and oil, and energy renewable resources.
There is not a place of mass disposal for ammonia, considering that most of the ammonia produced is used in products that we use in our own homes. The disposal of household ammonia products is not difficult if you know the proper procedures and cautions that should be taken. For instance, when taking into consideration that ammonia is a harmful and toxic chemical, it is actually relatively simple to dispose of it safely without contaminating other things. This process involves you simply adding a large quantity of water to it to dilute the toxicity of the product and then proceeding to dump it down a drain. By doing this, it allows you to avoid contaminating the soil or other natural resources and places with it.
Rather than have direct benefits towards Earth, the use of ammonia is more of a better alternative. It can be used as a replacement in refrigeration, and therefore reduce the direct greenhouse gas emissions. Also, its energy efficiency lowers indirect greenhouse gas emissions. As a “plus side” the use of ammonia doesn’t affect the ozone layer or contribute to global warming, so it is safe to say that its use isn’t necessarily “bad”.
To this day, ammonia is being produced and sold all over the world. Taking in the fact that it is used in products in our homes, its production obviously isn’t illegal. But, this isn’t to say that there aren’t regulations that need to be met. Ammonia producers have to have regular inspections to be sure that their equipment is safe and up to date, to make sure that their workers are properly trained so that they know what they are actually doing, and that the workers are also aware of what to do if there was to be an emergency involving the chemical. So while it isn’t illegal to produce ammonia, it is illegal for the companies to continue to produce it if they have not followed these requirements.
Ammonia is both caustic and hazardous. Despite how plants might believe they have things controlled, occurrences like the recent ammonia leak from an ice-making plant in Ningde City, China, can occur and produce life threatening possibilities. The people were awoken from their sleep by the pungent smell and sight of the ammonia leak. Soon after, an immediate evacuation of about 500 people took place, and several emergency teams were called in to secure the area and take care of the problem. Thankfully, everyone survived and few were admitted into the local hospital. So even though cases like this rarely occur, there are always risks in the production of ammonia.
Works Cited: Shakhashiri, Bassam Z. "Chemical of the Week." Online posting. Ammonia, NH3. Science is Fun in the Lab of Shakhashiri. Bassam L. Shakhashiri, Feb. 2008. Web. 1 Jan. 2010. <http://scifun.chem.wisc.edu/chemweek/pdf/ammonia.pdf>. The Internet Encyclopedia of Science. N.p, n.d. Web. 1 Jan. 2010. <http://www.daviddarling.info/encyclopedia/A/ammonia.html>. "Ammonia." Online posting. Ammonia: The Story Behind the Gas. N.p., n.d. Web. 1 Jan. 2010. <http://www.chm.bris.ac.uk/webprojects2001/prime/>. Clark, Jim.Online posting. Haber Process. Jim Clark, 2002. Web. 1 Jan. 2010. <http://www.chemguide.co.uk/physical/equilibria/haber.html>. "Ningde City, 500 ice-making plant emergency evacuation of ammonia leak." China Daily. N.p., 10 Nov. 2010. Web. 1 Jan. 2010. <http://www.china-daily.org/China-News/Ningde-City-500-ice-making-plant-emergency-evacuation-of-ammonia-leak/>. "Ammonia." Wikipedia: The Free Encyclopedia. Wikimedia Foundation, Inc. , 2010. Web. 1 Jan. 2010. <http://en.wikipedia.org/wiki/Ammonia>. Ammonia 21 everything natural . Shecco, 2009. Web. 1 Jan. 2010. <http://www.ammonia21.com/faq.php>. H2I. Canadian Hydrogen and Fuel Cell Association , 2010. Web. 1 Jan. 2010. <http://www.poweringnow.ca/how-do-we-produce-hydrogen>. "Nitrogen ." Periodic Table of the Elements. University of California , 2003. Web. 1 Jan. 2010.<http://periodic.lanl.gov/elements/7.html>. Web. 1 Jan. 2010. <http://www.rsc.org/images/feature%20dronsfield%20fritz%20haber_tcm18-85279.jpg>. Web. 1 Jan. 2010. <http://www.green-living-made-easy.com/image-files/greengarage11.jpghttp://www.green-living-made-easy.com/image-files/greengarage11.jpg>. Web. 1 Jan. 2010. <http://www.caslab.com/Chemical-Search/Chemical-Structure/7664-41-7.gif>. Web. 1 Jan. 2010. <http://www.china-daily.org/image/20101110232725.jpg>. Web. 1 Jan. 2010. <http://edutail.com/wp-content/uploads/2010/09/nitrogen-cycle.jpeg>.