Strongyloides
Morning Report Dec 14 th , 2009 Nicole Cullen. Strongyloides. What is Strongyloides?. Parasitic infection with a predilection for the intestines 2 most common and clinically relevant species are: Strongyloides stercoralis Strongyloides fuelleborni Limited to Africa and Papua New Guinea.

Strongyloides
E N D
Presentation Transcript
Morning Report Dec 14th, 2009 Nicole Cullen Strongyloides
What is Strongyloides? • Parasitic infection • with a predilection for the intestines • 2 most common and clinically relevant species are: • Strongyloides stercoralis • Strongyloides fuelleborni • Limited to Africa and Papua New Guinea
Epidemiology • Relatively uncommon in the US • BUT, endemic areas in the rural parts of the Southeastern states and the Appalachian mountain area • Certain pockets with prevalence 4% • Usually found in tropical and subtropical countries • Prevalence up to 40% in areas of West Africa, the Caribbean, Southeast Asia • Affects >100 million worldwide • No sexual or racial disparities. All age groups.
How Do You Get It? • Penetration of intact skin by filiariform larvae in the soil, or ingestion through contaminated food or water • Larvae enter the circulation • Lungs alveoli ascension up tracheobronchial tree swallowed molt in the small bowel and mature into adult female • Females enter the intestinal mucosa and produce several eggs daily through parthenogenesis (hatch during transit through the gut)
Clinical Presentation • Acute infection: • Lower extremity itching (mild erythematous maculopapular rash at the site of skin penetration) • Cough, dyspnea, wheezing • Low-grade fevers • Epigastric discomfort, n/v/d
Clinical Presentation • Chronic Infection • Can be completely asymptomatic • Abdominal pain that can be very vague, crampy, burning • Often worse after eating • Intermittent diarrhea • Can alternate with constipation • Occasional n/v • Weight loss (if heavy infestation) • Larva currens (“racing larva” – a recurrent maculopapular or serpiginous rash) • Usually begins perianally and extends up the buttocks, upper thighs, abdomen • Chronic urticaria
Clinical Presentation • Severe infection • Can be abrupt or insidious in onset • N/v/d, severe abdominal pain, distention • Cough, hemoptysis, dyspnea, wheezing, crackles • Stiff neck, headache, MS changes • If CNS involved • Fever/chills • Hematemesis, hematochezia • Rash (petechiae, purpura) over the trunk and proximal extremities • Caused by dermal blood vessel disruption brought on by massive migration of larvae within the skin • Risk factors for severe infection • Immunosuppressant meds (steroids, chemo, TNF modulators, tacro, etc – all BUT cyclosporine) • Malignancy • Malabsorptive state • ESRD • DM • Advanced age • HIV • HTLV1 • Etoh
Clinical Presentation • Can replicate in the host for decades with minimal or no sx • High morbidity and mortality when progresses to hyperinfection syndrome or disseminated strongyloidiasis • Usually in immunocompromised hosts (pregnancy?)
Dangerous Complications • Hyperinfection Syndrome • Acceleration of the normal life cycle, causing excessive worm burden • Autoinfection (turn into infective filariform larva within the lumen • Spread of larvae outside the usual migration pattern of GI tract and lungs • Disseminated strongyloidiasis • Widespread dissemination of larvae to extraintestinal organs • CNS (meningitis), heart, urinary tract, bacteremia, etc • Can be complicated by translocation of enteric bacteria • Travel on the larvae themselves or via intestinal ulcers • Mortality rate close to 80% • Due to delayed diagnosis, immunocompromised state of the host at this point
Laboratory Findings • CBC • WBC usually wnl for acute and chronic cases, can be elevated in severe cases • Eosinophilia common during acute infection, +/- in chronic infection (75%), usually absent in severe infection
Diagnostic Testing • Stool O&P • Microscopic ID of S. sterocoralis larvae is the definitive diagnosis • Ova usually not seen (only helminth to secrete larva in the feces) • Stool wet mount (direct exam) • In chronic infection, sensitivity only 30%, can increase to 75% if 3 consecutive stool exams • Can enhance larvae recovery with more obscure methods (Baermann funnel, agar plate, Harada-Mori filter paper)
Wet Mount Larva seen via direct examination of stool
Serology • ELISA • Most sensitive method (88-95%) • May be lower in immunocompromised patients • Cannot distinguish between past and present infections • Can cross-react with other nematode infections • If results are positive, can move on to try and establish a microscopic dx
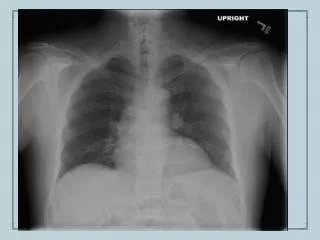
Imaging • CXR – patchy alveolar infiltrates, diffuse interstitial infiltrates, pleural effusions • AXR – Loops of dilated small bowel, ileus • Barium swallow – stenosis, ulceration, bowel dilitation • Small bowel follow-through – worms in the instestine • CT abdomen/pelvis – nonspecific thickening of the bowel wall
Procedures • EGD – duodenitis, edematous mucosa, white villi, erythema • Colonoscopy – colitis • Duodenal aspiration – examine for larvae • Sputum sample, bronchial washings, BAL – show larvae • Sputum cx • Nl respiratory flora organisms pushed to the outside in groups as a result of migrating larvae • Characteristic pattern can be diagnostic of S.Stercoralis infection • If CNS involved, LP – gram stain, cell count/diff ( protein, ↓ glu, poly predominance), wet mount prep
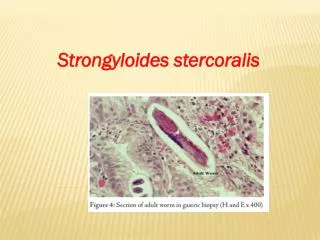
Histology • Larvae typically found in proximal portion of small intestine • Embedded in lamina propria • Cause edema, cellular infiltration, villous atrophy, ulcerations • In-long standing infections, may see fibrosis
Treatment • Antihelminitic therapy • Ivermectin • Albendazole • Thiabendazole • Abx directed toward enteric pathogens if bacteremia or meningitis (2-4wks) • Minimize immunosuppression as possible • Directed supportive tx • Transfusions if GI bleed, antihistamines for itching, surgery if bowel perf, etc • Repeat course of antihelminitic therapy if immunocompromised, as relapse common
Follow-Up • Repeat stool exams or duodenal aspirations in 2-3 mos to document cure • Repeat serologies 4-8 mos after therapy • Ab titer should be low or undetectable 6-18 mos after successful tx • If titer not falling, additional antihelminitic tx • Precautions for travelers to endemic areas, but no prophylaxis or vaccine available
References • Arch EL, Schaefer JT and Dahiya A. Cutaneous manifestation of disseminated strongyloidiasis in a patient coinfected with HTLV-1. Dermatology Online Journal. 2008;14(12):6. • Chadrasekar PH, Bharadwaj RA, Polenakovik H, Polenakovik S. Emedicine: Strongyloidiasis. April 3, 2009. • Concha R, Harrington W and Rogers A. Intestinal Strongyloidiasis. Recognition, Management and Determinants of Outcome. Journal of Clinical Gastroengerology. 2005;39(3):203-211. • Greiner K, Bettencourt J, and Semolic C. Strongyloidiasis: A Review and Update by Case Example. Clinical Laboratory Science. 2008;21(2):82-8. • Siddiqui AA, Berk SL. Diagnosis of Strongyloides stercoralis infection. Clin Infect Dis. October 1, 2001;33:1040-7. • Zeph, Bill. Strongyloides stercoralis Infection Can Be Fatal. American Family Physician.March 15, 2002.