Energetics
Energetics. IB. Exothermic and Endothermic. Chemical reactions involves the breaking and the making of bonds. Energy is needed to break down a bond. Energy is released when a bond is formed. If more energy is released than is absorbed, the reaction will be Exothermic.

Energetics
E N D
Presentation Transcript
Energetics IB
Exothermic and Endothermic • Chemical reactions involves the breaking and the making of bonds. • Energy is needed to break down a bond. • Energy is released when a bond is formed. • If more energy is released than is absorbed, the reaction will be Exothermic. • If more energy is needed to break the bonds than is given when new bonds are formed, the reaction will be endothermic.
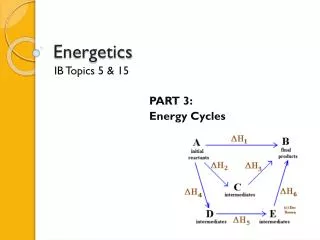
Activation energy Energy given out by reaction Using a catalyst might lower the activation energy Energy level diagrams Energy level Reaction progress
Enthalpy • The energy contained in a chemical bond that can be converted into heat is known as enthalpy. • Enthalpy is given the symbol H. • Enthalpy can not be measured directly, but we can measure the enthalpy change in a reaction, ΔH
Endothermic and Exothermic • A process is endothermic when H is positive.
Endothermic and Exothermic • A process is endothermic when H is positive. • A process is exothermic when H is negative.
ΔH sign • EXOTHERMIC – more energy is given out than is taken in (burning, respiration)
ENDOTHERMIC – energy is taken in but not necessarily given out. (photosynthesis)
Same magnitude; different signs. Reversing a Reaction • DH changes sign when a process is reversed. • Therefore, a cyclic process has the value DH = 0. Chapter 6: Thermochemistry
Using DH Values are measured experimentally Negative values indicate exothermic reactions Positive values indicate endothermic reactions Changes sign when a process is reversed. Therefore, a cyclic process has the value DH = 0 For problem-solving, one can view heat being absorbed in an endothermic reaction as being like a reactant and heat being evolved in an exothermic reaction as being like a product
Calorimetry • We measure heat flow using calorimetry. • A calorimeter is a device used to make this measurement. • A “coffee cup” calorimeter may be used for measuring heat involving solutions. A “bomb” calorimeter is used to find heat of combustion; the “bomb” contains oxygen and a sample of the material to be burned.
Calorimetry Relationships Specific heat of a substance is the amount of heat required to raise the temperature of one gram by 1 o C or by 1 Kelvin. Specific heat = C = q/mDT units of C: J g–1oC–1 or J g–1 K–1
Specific Heats EOS Chapter 6: Thermochemistry
Standard Enthalpies The standard enthalpy of reaction (DHo) is the enthalpy change for a reaction in which the reactants in their standard states yield products in their standard states
Enthalpies of Formation The standard enthalpy of formation (DHof) of a substance is the enthalpy change that occurs in the formation of 1 mol of the substance from its elements when both products and reactants are in their standard states
Enthalpies of Combustion • Is the enthalpy change that occurs during the complete combustion of one mole of a substance. • The enthalpy of combustion is defined in terms one mole of reactants, where the enthalpy of formation is defined in terms of one mole of products. • the symbol DHo Cis used to represent standard enthalpy change os combustion.
Standard Enthalpiesof Formation at 25 oC EOS Chapter 6: Thermochemistry
When multiplying equation coefficients, multiply values of DH for that reaction Hess’s Law ofConstant Heat Summation The heat of a reaction is constant, regardless of the number of steps in the process DHoverall = S DH’s of individual reactions When it is necessary to reverse a chemical equation, change the sign of DH for that reaction
Hess’s Law • H is well known for many reactions, and it is inconvenient to measure H for every reaction in which we are interested. • However, we can estimate H using H values that are published and the properties of enthalpy.
Enthalpies of Reaction The change in enthalpy, H, is the enthalpy of the products minus the enthalpy of the reactants: H = Hproducts−Hreactants
Hess’s Law Hess’s law states that “If a reaction is carried out in a series of steps, H for the overall reaction will be equal to the sum of the enthalpy changes for the individual steps.”
Calculation of H C3H8 (g) + 5 O2 (g) 3 CO2 (g) + 4 H2O (l) • Imagine this as occurring in 3 steps: C3H8 (g) 3 C(graphite) + 4 H2 (g) 3 C(graphite) + 3 O2 (g) 3 CO2 (g) 4 H2 (g) + 2 O2 (g) 4 H2O (l)
Calculation of H C3H8 (g) + 5 O2 (g) 3 CO2 (g) + 4 H2O (l) • The sum of these equations is: C3H8 (g) 3 C(graphite) + 4 H2 (g) 3 C(graphite) + 3 O2 (g) 3 CO2 (g) 4 H2 (g) + 2 O2 (g) 4 H2O (l) C3H8 (g) + 5 O2 (g) 3 CO2 (g) + 4 H2O(l)
Calculation of H We can use Hess’s law in this way: H = nHf(products) - mHf(reactants) where n and m are the stoichiometric coefficients.
Calculation of H C3H8 (g) + 5 O2 (g) 3 CO2 (g) + 4 H2O (l) H = [3(-393.5 kJ) + 4(-285.8 kJ)] - [1(-103.85 kJ) + 5(0 kJ)] = [(-1180.5 kJ) + (-1143.2 kJ)] - [(-103.85 kJ) + (0 kJ)] = (-2323.7 kJ) - (-103.85 kJ) = -2219.9 kJ