Chemical Elements and Water
360 likes | 610 Views
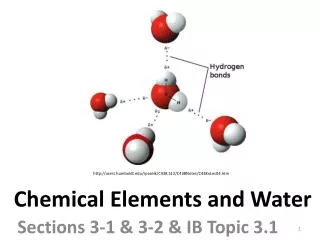
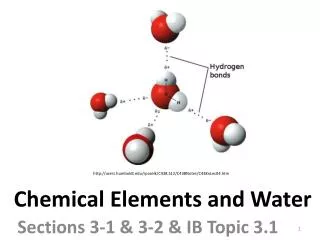
Chemical Elements and Water. Sections 3-1 & 3-2 & IB Topic 3.1. http://users.humboldt.edu/rpaselk/C438.S12/C438Notes/C438nLec04.htm. Many elements occur frequently in living organisms or are of use in metabolic processes. The most frequently occurring elements in living organisms are…?.

Chemical Elements and Water
E N D
Presentation Transcript
Chemical Elements and Water Sections 3-1 & 3-2 & IB Topic 3.1 http://users.humboldt.edu/rpaselk/C438.S12/C438Notes/C438nLec04.htm
Many elements occur frequently in living organisms or are of use in metabolic processes. The most frequently occurring elements in living organisms are…? http://i-biology.net/
Can you explain what these symbols are? What is this? What are these symbols?
Ions: An unreactive (stable) atom has a full outer electron shell. The inner shell is ‘full’ with two electrons. Subsequent shells are ‘full’ with eight electrons. Inner shell = 2 (full). Outer shell = 1 (not full – lose one!) Inner shell = 2 full outer shell = 7 (not full – gain one!)
Now: More electrons Than protons So negative ANION e- moves Now: More protons Than electrons So positive Cation http://www.footprints-science.co.uk/ionic.htm
Carbon and hydrogen are the foundation of organic molecules – molecules found in living things. The pH of a solution is a measure of the activity of dissolved H+ ions. A low pH (1- 6) signifies a high concentration of H+ ions. A high pH (8 – 14) signifies low H+ concentration.
A proton gradient in use. http://www.youtube.com/watch?v=U26Jz3K1w2k
Oxygen in respiration http://www.youtube.com/watch?v=xbJ0nbzt5Kw http://highered.mcgraw-hill.com/olc/dl/120071/bio11.swf
Oxygen is also used in oxidation reactions: Oxygen is put in. For example, breaking phosphate from ATP to release energy in tissues.
Important nitrogen compounds: • Nitrogen: N2 • Ammonium: NH4+ • Nitrates: NO3- • Nitrites: NO2- • Urea: (NH2)2CO http://www.biology.ualberta.ca/facilities/multimedia/uploads/ecology/ncycle.swf
Iron is an important micronutrient in living things. http://www.masimo.com/aboutmasimo/index.htm
Iron is often a limiting factor in plant productivity. Experiments of iron seeding can have dramatic effects, such as this phytoplankton bloom seen in the Baltic sea. http://phys.org/news69420332.html
Calcium is an essential mineral in many species. Horseshoe crab http://outreach.mcb.harvard.edu/animations/synaptic.swf
Phosphorus is essential in the formation of the phospholipid bilayer: Phosphates
Sulfur is found in some amino acids. The ‘R’ group Is the variable • It is also a reactant for chemosynthetic bacteria (chemoautotrophs) found in deep sea vents. These bacteria produce organic molecules from hydrogen sulphide, carbon dioxide and oxygen. Where does the CO2 and O2 come from? Even though light is not directly part of the process, does chemosynthesis still reply on sunlight to an extent?
A ‘black smoker’ hydrothermal vent http://extrememarine.org.uk/outofthisworldoctopus/5-hydrothermal-vents/
Blood plasma This is an example of active transport. Cell Cytoplasm http://chaitanya1.wordpress.com/2007/11/15/listening-to-the-body/sp-pump/
Let’s start out with this animation on the properties of water. http://www.sumanasinc.com/webcontent/animations/content/propertiesofwater/water.swf
Cohesive properties of water • A single hydrogen bond is not very strong. A large number of hydrogen bonds are very strong. Each water molecule bonds with four others in a tetrahedral arrangement.
Cohesive properties of water Can you do this? Cohesion forms droplets. Surface tension keeps them spherical. Adhesion sticks them to the leaf. http://i-biology.net/
Thermal properties of water Water has a high specific heat capacity. This means that it takes a lot of energy for the temperature of water to change states. This is because there are so many H-bonds. This all means that the temperature of water remains relatively stable. Most organisms are adapted to a narrow range of conditions. The slow heating and cooling of water are ideal for these organisms – there is less risk of extreme changes. Water moderates temperature changes.
Water as a coolant It takes a lot of energy for water to change temperature. This means that it will heat or cool more slowly than air or land. So animals can use mud or water to cool off on a hot day.
Water as a coolant Water makes up 70% of the body, including he blood. Because it is resistant to temperature change, cooler blood from some parts of the body can be circulated to other parts, cooling them down. http://creationwiki.org/Circulatory_system
Solvent properties of water http://www.northland.cc.mn.us/biology/Biology1111/animations/dissolve.swf Polar attractions cause water molecules to surround and isolate the solute molecules.
Solvent properties of water Click on the link to try out another animation! http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/molvie1.swf
Water as a medium for metabolic reactions: All metabolic reactions (reactions in living things) occur in solution meaning the reactants are dissolved. Click to See animation What is a metabolic reaction? http://iweb.tntech.edu/mcaprio/diffusion-animated.gif http://www.wisc-online.com/objects/MBY2604/MBY2604.swf
Water as a medium for metabolic reactions: http://www.3dscience.com/3D_Images/Human_Anatomy/Respiratory/Alveoli/Alveoli.php
Floating of Ice on Liquid Water • Ice floats in liquid water because hydrogen bonds in ice are more “ordered,” making ice less dense
Figure 3.6 Hydrogen bond Liquid water: Hydrogen bonds break and re-form Ice: Hydrogen bonds are stable
Water as a transport medium Water is a good solvent (yup.. still saying that!): it dissolves nutrients, gases, and waste products. These can be carried in the circulatory system of animals, through xylem and phloem in vascular plants or through the water in soil or aquatic habitats. Water can transport molecules across membranes in diffusion, as well as within the cell or interstitial (between-cell fluid). http://www.mhhe.com/biosci/esp/2001_gbio/folder_structure/ce/m3/s2/index.htm http://rdouglasfields.wordpress.com/page/2/