Electrochemistry
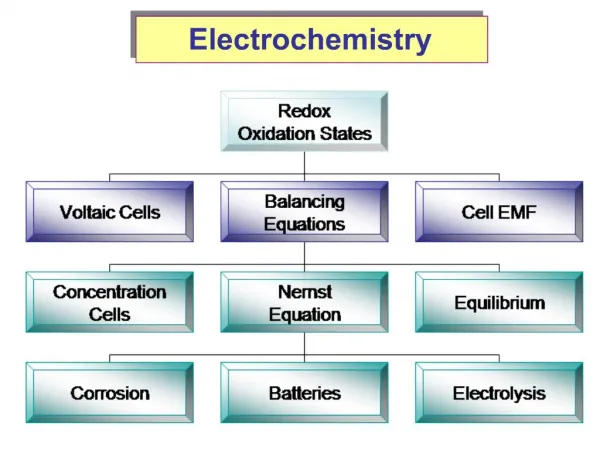
Electrochemistry. The study of the exchange of chemical and electrical energy. Oxidation-Reduction reactions take place in electrochemical cells. Each electrochemical cell consists of two solid conductors called electrodes. Two types of electrochemical cells: Voltaic Cell Electrolytic Cell.

Electrochemistry
E N D
Presentation Transcript
The study of the exchange of chemical and electrical energy. • Oxidation-Reduction reactions take place in electrochemical cells. • Each electrochemical cell consists of two solid conductors called electrodes.
Two types of electrochemical cells: • Voltaic Cell • Electrolytic Cell
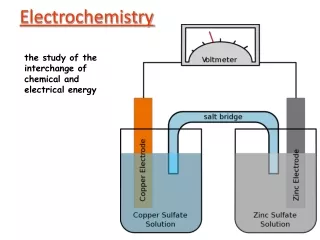
Voltaic Cell • Reactions are spontaneous (Table J) • Redox reactions produce electrical energy • Lets look at an example: Cu+2(aq) + Zn (s) -------> Cu (s) + Zn+2(aq)
Each Voltaic cell is composed of two half cells. Zinc electrode Copper electrode
The anode, is the electrode where the oxidation of zinc occurs: Zn (s) ----------> Zn+2(aq) + 2e- As zinc is oxidized, the zinc electrode will slowly dissolve to produce zinc ions, which enter into the solution. Anode (negative)
The cathode is the electrode where the reduction of copper occurs: Cu+2(aq) + 2e- -------> Cu (s) As copper is reduced, copper atoms accumulate on the surface of the solid copper electrode. Cathode (positive)
Looking at the two cells, can a redox reaction take place? The reaction will not occur.There is no way for the electrons lost by Zinc to get over to the Copper ions and reduce them to Copper metal.
What if we provide a path for the electrons released by the oxidation of the zinc to get over to the copper? Now that the electrons have a path to the Cu/Cu2+ side it would appear that the reaction can proceed, Why? but it does not.
As the redox reaction proceeds: • There is a build up of Zn2+ ions in the solution where the Zinc is being oxidized • At the same time, Cu2+ ions are removed from solution where the Copper ion is being reduced • As a result, there is a build up of a positive charge in the zinc solution, and a negative charge in the copper solution. These charges do not allow electrons to flow. • The positive charge in the zinc solution makes it harder for the negative electrons to leave and the negative charge in the copper solution repels the electrons that are trying to come over from the zinc side We need a way to neutralize the charge build-up in the solutions due to the change in ion concentration
What if we had a tube filled with aqueous solution that connected the two redox reactions? Electron flow This tube contains an electrolyte and is called a salt bridge.
The salt bridge allows the electrons to flow, keeping the overall concentration of ions in balance between the two sides. • By connecting the salt bridge we have movement of both types of ions: • anions are going into the oxidation side and • cations are going into the reduction side
Standard Cell potential • Electrical energy produced by a redox reaction is measured as voltage • Some standard electrode potentials are listed below. Electrode Process Eo/V Li+(aq) + e- Li(s) -3.03 K+(aq) + e- K(s) -2.92 Zn2+(aq) + 2e- Zn(s) -0.76 Cu2+(aq) + 2e- Cu(s) +0.34
Electrolytic Cell • Reactions are non spontaneous • Redox reactions require electrical energy to occur
Anions are attracted towards the anode where they undergo oxidation. Electrons flow from the anode to the cathode where cations undergo reduction.
Use of Electrochemical Cell • A common dry cell battery.
Use of Electrolytic Cell • Electroplating • Electrolysis of water