Equilibrium Reaction
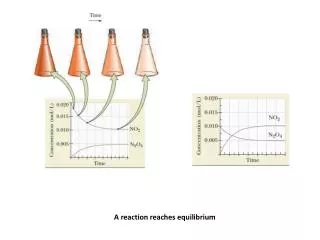
Equilibrium Reaction. Chemical equilibrium is the state reached by a reaction mixture when the rates of forward and reverse reactions become equal . Equilibrium is dynamic. A Reaction at Equilibrium. Equilibrium Constant , K c.

Equilibrium Reaction
E N D
Presentation Transcript
Equilibrium Reaction Chemical equilibrium is the state reached by a reaction mixture when the rates of forward and reverse reactions become equal. Equilibrium is dynamic
Equilibrium Constant, Kc Since the equilibrium concentrations do not change with time, a relation can be established between them. This is known as the law of mass action says that the equilibrium constant of any chemical reaction obeys the above expression whatever the equilibrium concentrations are substituted into the expression, at constant temperature.
Equilibrium Constant, KP If all reactants and products are gases, another equilibrium constant, KP, can be written.