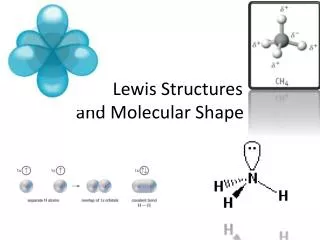
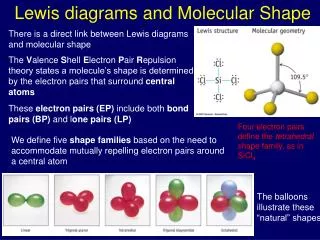
4.6 Molecular Shape
4.6 Molecular Shape. To determine the shape around a given atom, first determine how many groups surround the atom. A group is either an atom or a lone pair of electrons. Use the VSEPR theory to determine the shape. The most stable arrangement keeps the groups

4.6 Molecular Shape
E N D
Presentation Transcript
4.6 Molecular Shape • To determine the shape around a given atom, • first determine how many groups surround the • atom. • A group is either an atom or a lone pair of • electrons. • Use the VSEPR theory to determine the shape. • The most stable arrangement keeps the groups • as far away from each other as possible.
VSEPR theory • VSEPR • (V ) Valence • (S) Shell • (E) Electron • (P) Pair • (R) Repulsion
VSEPR theory The idea of VSEPR is that the valence electron pairs surrounding an atom mutually repel each other, and will therefore adopt an arrangement that minimizes this repulsion, thus determining the molecular geometry.
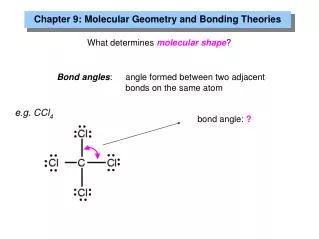
Two Groups Around an Atom • Any atom surrounded by only two groups is linearand has a bond angle of 180o. • An example is CO2: • Ignore multiple bonds in predicting geometry.Count only atoms and lone pairs.
Three Groups Around an Atom • Any atom surrounded by three groups is • trigonal planar and has bond angles of 120o. • An example is H2CO:
Four Groups Around an Atom • Any atom surrounded by four groups is • tetrahedral and has bond angles of 109.5o. • An example is CH4:
Four Groups Around an Atom • If the four groups around the atom include one • lone pair, the geometry is a trigonal pyramid • with bond angles of ~109.5o. • An example is NH3:
Four Groups Around an Atom • If the four groups around the atom include two • lone pairs, the geometry is bent and the bond • angle is 105o (i.e., close to 109.5o). • An example is H2O:
Question 4.65 • Give the molecular shape around the boron atom in BCl3 andthe nitrogen atom in NCl3 and explain why they are different. • BCl3is trigonal planar because it has three Cl’s bonded to B but no lone pairs. NCl3 is trigonal pyramidal because it has three Cl’s around N as well as a lone pair.
4.7 Electronegativity and Bond Polarity • Electronegativity is a measure of an atom’s • attraction for e− in a bond. • It tells how much a particular atom “wants”e−.
Electronegativity and Bond Polarity • If the electronegativities of two bonded atoms • are equal or similar, the bond is nonpolar. • The electronsin the bond are being shared equallybetween the two atoms.
Electronegativity and Bond Polarity • Bonding between atoms with different electro- • negativities yields a polar covalent bond or dipole. • The electrons in the bond are unequally shared • between the C and the O. • e− are pulled toward O, the more electronegative • element; this is indicated by the symbol δ−. • e− are pulled away from C, the less electronegative • element; this is indicated by the symbolδ+.
Question 4.75 • Lable the bond formed between carbon and each of the following elements are nonpolar, polar or ionic. • Calculate the electronegativity difference between the atoms and carbon and use the following rules: • less than 0.5 (nonpolar); 0.5–1.9 (polar covalent); greater than 1.9 (ionic). • a. C = (2.5 – 2.5) = 0 • nonpolar • b. O = (3.5 – 2.5) = 1.0 • Polar • c. Li = (2.5 – 1.0) = 1.5 • Polar • d. H = (2.5 – 2.1) = 0.4 • nonpolar
Question 4.78 and 4.80 • Which bond in each pair is more polar, that is has more electronegativity difference between atoms then label each bond with to show direction of polarity • Si–O = (3.5 – 1.8) = 1.7 • Si–S = (2.5 – 1.8) = 0.7 • b. H–F = (4.0 – 2.1) = 1.9 • H–Br = (2.8 – 2.1) = 0.7 • c. C–B = (2.5 – 2.0) = 0.5 • C–Li = (2.5 – 1.0) = 1.5 more polar more polar more polar
4.8 Polarity of Molecules The classification of a molecule as polar or nonpolar depends on: • The polarity of the individual bonds • The overall shape of the molecule Nonpolar molecules generally have: • No polar bonds • Individual bond dipoles that cancel Polar molecules generally have: • Only one polar bond • Individual bond dipoles that do not cancel