The Mole
The Mole The Mole It's not a spy, a machine for digging tunnels, a burrowing animal, or a spot of skin pigmentation - it's - a unit of measurement containing about 6.02 x 10 23 particles. Chemists created the mole as their own counting unit. The Chemical Mole 1mole of atoms is

The Mole
E N D
Presentation Transcript
It's not a spy, a machine for digging tunnels, a burrowing animal, or a spot of skin pigmentation - it's - a unit of measurement containing about 6.02 x 1023 particles. • Chemists created the moleas their own counting unit. The Chemical Mole
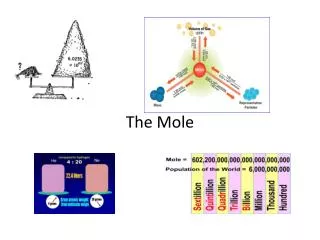
1mole of atoms is 602,214,199,000,000,000,000,000 atoms!
Avogadro’s Number 6.02 x 1023 particles / mole
Just How Big is a Mole? • Enough soft drink cans to cover the surface of the earth to a depth of over 200 miles. • If you had Avogadro's number of unpopped popcorn kernels, and spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. • If we were able to count atoms at the rate of 10 million per second, it would take about 2 billion years to count the atoms in one mole.
Moles of Elements • 1 mole element = (atomic mass) in grams of that element 1 mole C = 12.011 g C 1 mole B = 10.81 g B 1 mole Cu = 63.55 g Cu
Avogadro’s Number as Conversion Factor 6.02 x 1023 particles 1 mole or 1 mole 6.02 x 1023 particles Note that a particle could be an atom OR a molecule!
Equation to find number of particles! 6.02 x 1023 particles 1 mole number of moles x = number of particles Example: How many particles of sucrose are in 3.50 moles? 6.02 x 1023 molecules 1 mole 3.50 mol sucrose x = 2.11 x 1024 molecules of sucrose
Equation to find number of moles! number of particles x = number of moles 1 mole 6.02 x 1023 particles Example: How many moles are contained in 4.50 x 1024 atoms of zinc? 1 mole Zn 6.02 x 1023 particles 4.50 x 1024 atoms of zinc x = 7.48 mol Zn
Learning Check 1. Number of atoms in 0.500 mole of Al a) 500 Al atoms b) 6.02 x 1023 Al atoms c) 3.01 x 1023 Alatoms 2.Number of moles of S in 1.8 x 1024 S atoms a) 1.0 mole S atoms b) 3.0 mole S atoms c) 1.1 x 1048 mole S atoms
Molar Mass 11.2
Molar Mass • The Mass of 1 mole (in grams) • Molar mass is equal to the atomic mass (look on the periodic table) 1 mole of C atoms = 12.01 g 1 mole of Au atoms = 196.97g 1 mole of Cu atoms = 63.55 g • Always round to the hundredths place.
Molar Mass of Molecules and Compounds Mass in grams of 1 mole equal numerically to the sum of the atomic masses 1 mole of CaCl2 Ca = 40.08 Cl = 35.453 x 2 70.906 40.08 + 70.906 = 110.99 g/mol
Molar Mass of Molecules and Compounds 1 mole of N2O4 N = 14.007 O = 15.999 x 2 x 4 28.014 63.996 28.014 + 63.996 = 92.01 g/mol
Where will you find the number of grams per mole? Mole to Mass Conversion Number of moles x number of grams = mass 1 mole Example: Calculate the mass in grams of 0.0450 moles of chromium. 0.0450 moles Cr x 52.0 g Cr = 2.34 g Cr 1 mol Cr The Periodic Table Look at your Periodic Table
Where will you find the number of grams per mole? Mass to Mole Conversion Mass x 1 mole = number of moles number of grams Example: How many moles of calcium are there in 525 grams of calcium? 525 g Ca x 1 mol Ca = 13.1 mol Ca 40.1 g Ca The Periodic Table Look at your Periodic Table
Mass to Atoms Conversion Two steps: • Use the mass to moles conversion • Now calculate the number of atoms using the molesto particlesequation.
Example: How many atoms of gold are in a pure gold nugget having a mass of 25.0 g? Step 1 Mass x 1 mole = number of moles number of grams 25.0 g Au x 1 mol Au = 0.13 mol Au 197.0 g Au Round to the hundredths place!!!!!!
6.02 x 1023 particles 1 mole number of moles x = number of particles Step 2 0.13 mol Au x = 7.83 x 1022 atoms Au 6.02 x 1023 atoms 1 mole Round to the hundredths place!!!!!!
Atoms/Molecules and Grams How many atoms of Cu are present in 35.4 g of Cu? 35.4 g Cu 1 mol Cu 6.02 X 1023 atoms Cu 63.5 g Cu 1 mol Cu = 3.4 X 1023 atoms Cu
Learning Check! How many atoms of K are present in 78.4 g of K? 78.4 g K 1 mol K 6.02 X 1023 atoms K 39.1 g K 1 mol K = 1.20 X 1024 atoms K
Percent Composition Mass of element x 100 = percent by mass Mass of compound Example: Determine the percent composition of NaHCO3. Determine the mass of each element first! %mass element = mass of element in 1 mol compound x 100 molar mass of compound Percent Na = 22.99 g Na x 100 = 27.37% Na 84.01 g NaHCO3 You have to complete this step for each element in the compound!!!!
Determine the Percent Composition from the Formula C2H5OH • Divide the mass of each element by the molar mass of the compound and multiply by 100%
Types of Formulas • Empirical Formula The formula of a compound that expresses the smallest whole number ratio of the atoms present. • Molecular Formula The formula that states the actual number of each kind of atom found in one molecule of the compound.
Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen • Convert the percentages to grams by assuming you have 100 g of the compound • Step can be skipped if given masses
Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen • Convert the grams to moles
Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen • Divide each by the smallest number of moles
Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen • If any of the ratios is not a whole number, multiply all the ratios by a factor to make it a whole number • If ratio is ?.5 then multiply by 2; if ?.33 or ?.67 then multiply by 3; if ?.25 or ?.75 then multiply by 4 Multiply all the Ratios by 3 Because C is 1.3
Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen • Use the ratios as the subscripts in the empirical formula C4H6O3
Molecular Formulas • The molecular formula is a multiple of the empirical formula • To determine the molecular formula you need to know the empirical formula and the molar mass of the compound
Determine the Molecular Formula of Benzopyrene if it has a molar mass of 252 g and an empirical formula of C5H3 • Divide the given molar mass of the compound by the molar mass of the empirical formula • Round to the nearest whole number
Determine the Molecular Formula of Benzopyrene if it has a molar mass of 252 g and an empirical formula of C5H3 • Multiply the empirical formula by the calculated factor to give the molecular formula • (C5H3)4 = C20H12