Proteins
Physical Therapy Faculty. Proteins. Biochemistry of . Outcomes:. By the end of the lecture, the students have the ability to. Recognize the structure of proteins and known examples for each. Recognize the importance of proteins in our life.

Proteins
E N D
Presentation Transcript
Physical Therapy Faculty Proteins Biochemistry of
Outcomes: By the end of the lecture, the students have the ability to • Recognize the structure of proteins and known examples for each. • Recognize the importance of proteins in our life. • Differentiate between variousclasses of amino acids. • Understand the mechanism and the different between hydrolysis and denaturation process and their biological applications. • Physical and chemical properties of proteins.
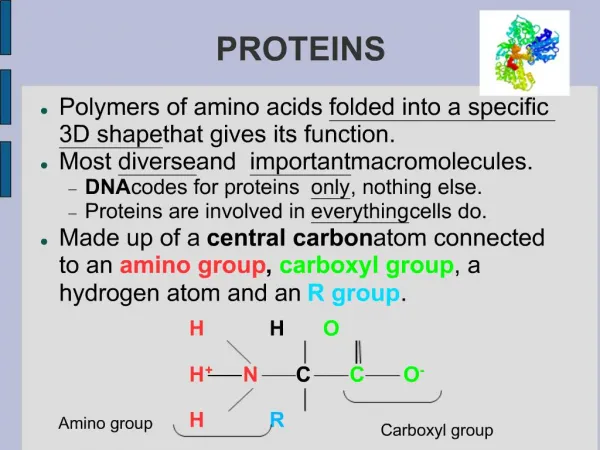
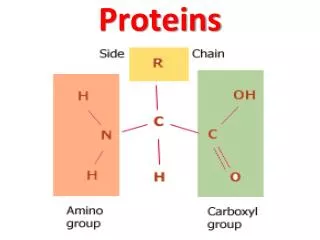
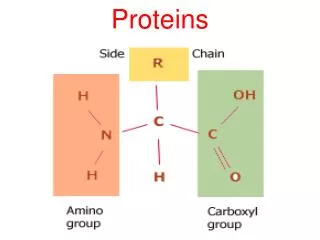
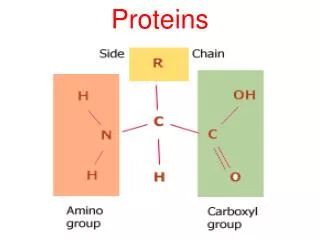
Protein structure and function • The primary structure of proteins It describes the order of the amino acids joined together to make the protein. A protein chain will have somewhere in the range of 50 to 2000 amino acid residues. The end of the peptide chain with the -NH2 group is known as the N-terminal, and the end with the -COOH group is the C-terminal. The "R" groups come from the 20 amino acids which occur in proteins. The peptide chain is known as the backbone, and the "R" groups are known as side chains.
The secondary structure of proteins Within the long protein chains there are regions in which the chains are organised into regular structures known as; alpha-helixes and beta-pleated sheets. These are the secondary structures in proteins. These secondary structures are held together by hydrogen bonds. These form between one of the lone pairs on an oxygen atom and the hydrogen attached to a nitrogen atom. The alpha-helix In an alpha-helix, the protein chain is coiled like a loosely-coiled spring. The "alpha" means that if you look down the length of the spring, the coiling is happening in a clockwise direction as it goes away from you. Beta-pleated sheets In a beta-pleated sheet, the chains are folded so that they lie alongside each other as an "anti-parallel" sheet.
Secondary structures of proteins are divided into one of two three dimensional forms, either alpha-helix or beta-pleated sheet. This is because hydrogen of the –NH group has a positive charge while the O of the –C=O group has a negative charge, so they form hydrogen bonds, causing polypeptide chains to twist. In the alpha helix the polypeptide chain is coiled into a cylindrical shape. Alpha-Helix In Beta-pleated sheet the polypeptide chains are linked in a parallel flat sheet.
The tertiary structure of proteins Alpha helices and beta pleated sheets can be twisted or moved into different and more complex forms. These are called tertiary structures and are the result of four different bonds: Ionic interactions Some amino acids (such as aspartic acid and glutamic acid) contain an extra -COOH group. Some amino acids (such as lysine) contain an extra -NH2 group. Hydrogen bonds Lots of amino acids contain groups in the side chains which have a hydrogen atom, hydrogen bonds between side groups - not between groups actually in the backbone of the chain, attached to either an oxygen or a nitrogen atom. van der Waals dispersion forces Several amino acids have quite large hydrocarbon groups in their side chains. Temporary fluctuating dipoles in one of these groups could induce opposite dipoles in another group on a nearby folded chain. Disulphide bond It is Sulphur bridges forming between two cysteine residues.
Quaternary structure It is the interaction between several chains of peptide bonds (subunits). The individual subunits are usually not covalently connected, but might be connected by a disulfide bond. Not all proteins have quaternary structure, since they might be functional as monomers. The quaternary structure is stabilized by the same range of interactions as the tertiary structure. Complexes of two or more polypeptides (i.e. multiple subunits) are called multimers. - a dimer if it contains two subunits - a trimer if it contains three subunits - a tetramer if it contains four subunits. Multimers made up of identical subunits may be referred to with a prefix of "homo-" (e.g. a homotetramer) and those made up of different subunits may be referred to with a prefix of "hetero-" (e.g. a heterotetramer, such as the two alpha and two beta chains of hemoglobin).
Examples of globular proteins Three-dimensional structure of myoglobin Myoglobin is a protein of 153 amino acids that is involved in oxygen transport. The polypeptide chain is folded around a heme group that serves as the oxygen-binding site. Hemoglobin is the iron-containing oxygen-transport metalloprotein in the red blood cells of vertebrates and the tissues of some invertebrates. In adult humans, the most common hemoglobin type is a tetramer (which contains 4 subunit proteins) called hemoglobin A, consisting of two α and two β subunits non-covalently bound, each made of 141 and 146 amino acid residues, respectively. This is denoted as α2β2
Proteins are classified by structure Shape Function • Simple • Conjugated • Glycoprotein: Immunogloulin. • Metalloprotein: Hemoglobin and Calmodulin • Nucleoprotein: RNA bound protein • Phosphoprotein:casein • Lipoprotein: Low density lipoprotein (LDL), HDL. • Globular • Fibrous 1. Catalytic proteins as enzyme 2. Structural proteins as collagen, keratin 3. Proteins serve as storage of nutrition as ferritin (store ion) or carrier as albumin. 4. Protective proteins as immunoglobulins 5. Regulatory proteins include hormones as insulin , glucagon,------etc. 6. proteins serve in Communication as neurotransmitters 7. proteins involve in motion as actin/myosin; in muscle 8. Transporter proteins as hemoglobin, transferrin.
Fibrous proteins water insoluble and found as structural materials, e.g. collagen, keratin, myosin. Globular proteins are compact, roughly spherical, water soluble and comprise all other types of protein as albumins and globulins.
Hydrolysis: Hydrolysis of proteins results in breaking down the peptide bonds to give amino acids thus it disrupts the primary structure of protein . Hydrolysis can be achieved by 1- Enzyme as Proteases. 2- Acids and bases Biological role of hydrolysis: 1-Convert inactive native prohormone into active hormone. ex; Proinsulin (inactive) → Insulin (active). Digestion of protein by enzyme as trypsin and pepsin.
Denaturation It is a process in which proteins can lose their structures and function, without breaking the peptide bonds bydenaturing agents as the following • Heat, U.V.,microwave radiation. • Heavy metal as mercury. • Soaps. • Organic acids as acetic acid. • Strong acids and bases as sulfuric acid and sodium hydroxide. Note: Denaturation disrupts 2ry,3ry,4ry structure of protein not 1ry structure.
1- Hemoglobin is a-------------protein. a-Monomeric. b-Hommultimeric. C-Heteromultimeric. • 2-Immunoglbulin is a----------protein. a-Regulatory. b-Protective. c-Structural. Choose: 3--------------is a biological catalyst that accelerate the biological reaction at physiological temperature. a-Hormone. b-Enzyme. c-Hemoglobin.
Correct the incorrect word: • Fibrous proteins are filamentous, water soluble and found as structural materials as myosin. • Denaturation disrupts 1ry structure. • Α-Helix and β-sheet are two forms of tertiary structure.
Complete: • Hemoglobin belongs to----------proteins • ---------------is a protein conjugate with nucleic acids. • LDL and HDL belong to--------------------. • Immunoglobin (IgG) is a ---------------proteins help in defense mechanism against virus and bacteria. • Ammonium sulfate causes protein precipitation by increasing ---------------. • ------------,----------,---------------are denaturating agents. • --------and--------are two isomers of the amide group.
Mention importance of: • Proteins in cell structure. • Proteins Hydrolysis. • Explain • Fibrous proteins more susceptible to hydrolysis than globular proteins. • Temperature affects on physical and chemical properties of proteins.