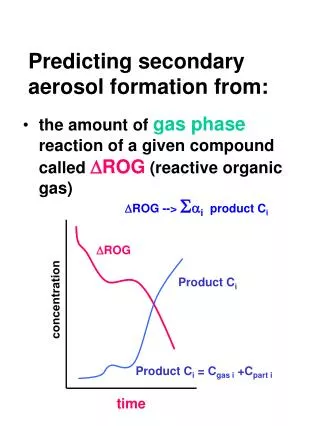
The relative importance of precursors to secondary aerosol formation will depend on: overall aerosol potential atmospher
What are the precursor compounds for secondary organic aerosols? What are the types of vegetation, vehicle exhaust, and burning that emit these precursors and under what conditions? R.Kamens, M. Jang, S. Lee, M. Jaoui, Depart. of Environ. Sci. and Eng. UNC-Chapel Hill kamens@unc.edu.

The relative importance of precursors to secondary aerosol formation will depend on: overall aerosol potential atmospher
E N D
Presentation Transcript
What are the precursor compounds for secondary organic aerosols? What are the types of vegetation, vehicle exhaust, and burning that emit these precursors and under what conditions? R.Kamens, M. Jang, S. Lee, M. Jaoui, Depart. of Environ. Sci. and Eng.UNC-Chapel Hill kamens@unc.edu
Secondary organic aerosol (SOA) material may be defined as organic compounds that reside in the aerosol phase as a function of atmospheric reactions that occur in either the gas or particle phases.
The relative importance of precursors to secondary aerosol formation will depend on: • overall aerosol potential • atmospheric emissions • presence of other initiating reactants (O3, OH, NO3, sunlight, acid catalysts)
1. Terpenoid 2. Aromatic 3. Particle Phase Reactions (aldehydes and alcohols)
Leonardo Da Vinvidescribed blue haze and thought that plant emissions were its source…(Went, 1959) Da Vinvi believed that it was due to water moisture emitted from the plants
F.W.Went published papers on biogenic emissions from vegetation over 40 years ago. He posed the question, “what happens to 17.5x107 tons of terpene-like hydrocarbons or slightly oxygenated hydrocarbons once they are in the atmosphere?” Went suggested that terpenes are removed from the atmosphere by reaction with ozone and demonstrated “blue haze” formation by adding crushed pine or fir needles to a jar with dilute ozone.
Different Terpene structures a-pinene b-pinene myrcene d-limonene
Synthesis of Terpenes From CO2 Ruzika, 1953 No mechanism for isoprene storage While terpenes can stored in resin duct
Global VOC Emissions Rates Estimates: Guenther et al, 1995 (Tg/y) Isoprene Monoterpenes ORVOC Total VOC Woods 372 95 177 821 Crops 34 6 45 120 Shrub 103 25 33 194 Ocean 0 0 2.5 5 Other 4 1 2 9 All 503 127 260 1150
Yu et al. Hannel et al a-pinene 22-119 36-148 b-pinene 16-119 7- 28** limonene 13- 63 0- 21 D3-carene 2- 21 8- 48 camphene 2- 21 5- 35 sabinene 0- 43 isoprene 0-228 Ambient Concentrations of selected terpenes (pptV)
Aerosol concentrations of selected terpenes products (ng m-3) 1ng m-3 =~0.1pptV Yu et al.Kavouras et al, 1998 Pinic acid 0.5 0.4- 85 pinonic acid 0.8 9 - 141 norpinonic acid 0.1- 38 Pinonaldehyde 1.0 0.2- 32 hydroxy-pinonaldehydes 0.5 oxo-liminoic acid 0.8 Nopinone 133 0.0 - 13
Sesquiterpenes (C15H24) There is a dearth of data on the emissions strength of sesquesterpenes compared to terpenes May contribute as much as 9% to the total biogenic emissions from plants. (Helmig ,et al, 1994) Flux data, Atlanta forest, Helmig et al., 1999
OH NO3 O3 a-Cedrene 2.6 hours 4 min 14 hours a-copaene 1.9 hours 2 min 2.5 hours b-Caryphyllene 53 min 2 min 2 min a-Humulene 36 min 1 min 2 min Longifiolene 3.7 hours 49 min 23 days Lifetimes of Sesquiterpenes average OH concentration =1.6x106; NO3 = 5x108 for 12 hours of night time; O3= 7x1011 (molecules cm-3)
Limonene Fluxes computed with and w/o an ozone scrubber (~50 ppb of O3 w/o O3 scrubber) over Fuentes, et al. 2000) Caryophylene with w/o
alcohols ketones alkanes p-cymen-8-ol* 2-heptanone n-hexane and C10-C17 cis-3-hexen-1-ol 2-methyl-6-methylene-1-7-octadien-3-one* Aromatics p-cymene linalool pinacarvone* alkenes acetates verbenone* 1-decene bornylacetate ethers 1-dodecene butylacetate* 1-,8 cinole 1-hexadecene* cis-3-hexenylacetate p-dimethylhydroxy benzene p-mentha-1,2,8-triene* aldehydes esters 1-pentadecene* n-hexanal methylsalicyclate* 1-tetradecene trans-2-hexenal Other emissions (Winer et al. , Kesselmeier and Staudt )
Factors that influence emissions 1. Temperature 2. light 3. injury
b-pinene emission rates per gram of dry biomass as a function of temperature (Fuentes, et al. 2000) E = Es exp {b (T-Ts)} Tingy et al.
a-pinene emissions compared to temp, and CO2 exchange (Mediterranean Oak,Kesselmeire et al ) a-pinene temp CO2 exchange
Changes in relative humidity were generally not deemed to be an important factor affecting terpene emissions (Guenther, JGR,1991)A young orange tree was exposed to drought stress by withholding water. Emissions of b-caryophyllene and trans-b-ocimene decreased little (-6%) from the non-drought conditions. Hansen and Seufert,(1999).
Emissions from drought-stressed apple leaves seem to show significant increases in hexanal, 2-hexenal, and hexanol (Ebel et al. 1995)Shade,et al (G. Res. Let.,1999) measured increases in monoterpene emissions of D-3 carene over a ponderosa pine plantation in the Sierra Nevada mountains after rain events and under high humidity, Tingey equation is corrected by multiplying by a relative humidity factor, BET.BET= cxRHn)/((1-cRHn)x(1+(c-1)xRHn) where c a constant, and RHna normalized relative humidity = (%relative humididy-18)/82
Plant damage Emissions from damaged leaves contain C6-aldehydes and alcohols.Temporary increases in terpene emissions have been observed from mounting plants in chambers.Isoprene emissions seem unaffected by plant damage. Injury to the bark of pine trees increases terpene emissions. Fungal attack on lodgepole pines releases terpenes and high amounts of ethanol, thought to attract pine beetles.
a-pinene Aerosol potential (Odum theory)
0.95 ppm a-pinene + 0. 44ppm NOx model data NO O3 NO2 NO2
Measured particle mass vs. model reacted a-pinene data model
O O particle phase pinonaldehye data model
a-pinene Aerosol potential (Odum theory)
a1 a2 Kom,1 Kom,2 %Yield (Y) D3-carene 0.057 .0476 0.063 0.0042 2 -11 caryophyllene 1.00 N/A 0.0416 N/A 17-64 a-humulene 1.00 N/A 0.0501 N/A 20-67 limonene 0.0239 0.363 0.055 0.0053 6 -23 a-pinene 0.038 0.326 0.171 0.0040 2- 8 b-pinene 0.113 0.239 0/094 0.0051 4-13 Griffin et al. biogenic aerosol yields
Relative aerosol potential of terpenoids Andersson-Sköld and Simpson, JGR, 2001
Griffin et al, JGR, 2000 Used a global photochemcial model to estimate the amount of terpenes and other biogenics that are reacted, DROGi.These were used in conjunction with specific compound “Odum fitting” constants to estimate total boigenic aerosol production on a yearly basis.This may be a conservative estimate because the fitting contents are derived at 308K, does not consider other aerosol surfaces, or particle phase reactions
Sienfeld and Pandis from from Kiehl, and Rodhe Natural emissions Tg /y anthropogenic Tg /yr Soil/mineral aerosol © 1500 Industrial dust © 100 Sea salt © 1300 Soot 10 Volcanic dust © 30 Sulfate from SO2 190 biological debris © 50 Biomass burning 90 Sulfates from biological gases 130 Nitrates from NOx 50 Volcanic Sulfates 20 VOCs 10 Nitrates 60 Biogenic aerosols 13-24 Total 3100 Total 450
Aromatics Globally, about 25 Tg/yr of toluene and benzene and are emitted with fossil fuels contributing ~80%, and biomass burning another 20 % (Ehhalt, 1999) A reasonable total aromatic emission rate might be 3 times the toluene+benzene emission rate.
Aromatics Volatile aromatic compounds comprise up to 45% in urban of the VOCs US and European locations. At rural sites it is 1-2% Toluene, m-and p-xylenes, benzene, 1,2,4-trimethyl benzene, o-xylene and ethylbenzene make up 60-75% of this load
Aromatics Tunnel studies show that aromatic emissions comprise 40-48% of the total nonmethane hydrocarbon emissions for LD and HD vehicles (Sagebiel, and Zielinska et al.) On a per mile basis heavy duty trucks emit more than twice the aromatic mass that light duty vehicles emit The same aromatics as found in ambient air, comprise 60% of the LD aromatic emissions and 27% of the HD
Aerosols from Aromatics (Chamber studies) 1. Odum et al. 2. Izumi et al. 3. Holes, et al. 4. Kliendienst et al. 5. Forstner et al. 6. Hurley et al 7. Jang and Kamens m-xylene
Particle phase reactions In UNC chamber experiments partitioning “Pankow” coefficients for aldehydes are much higher than predicted partitioning coefficients, calculated from the vapor pressures and activity coefficients (Jang and Kamens, ES&T, 2001, Kamens and Jaoui, ES&T, 2001 )
Pred log iKp Exp. log iKp 4.25 -3.8 5.64 -5.69 5.41 -3.86 6.10 -2.66 5.56 -3.23 5.38 -3.91 Toluene gas phase reaction reactions iKp i= 760 RTx10-6 fom /{Mw gi PoLi} exp iKp = [iCpart]/[iCgas xTSP]
Particle phase reactions Ziemann and Tobias have reported the formation of hemiacetals in the particle phase of secondary organic aerosols
Aldehyde functional groups can react in the aerosol phase through heterogeneous reactions viahydration, polymerization, and hemiacetal/acetal formation with alcohols. • Aldehyde reactions can be radically accelerated by acid catalysts such as particle sulfuric acid (Jang and Kamens, ES&T, 2001)
Why don’t we see these large highly oxygenated compounds?? Reverse reactions to the original aldehyde parent structures can occur during sample work up/solvent extraction procedures;
nebulizer (NH4)2SO4 Solution (NH4)2SO4+H2SO4 Solution aldehydes alcohols glyoxal aldehydes alcohols glyoxal 500 liter Teflon bag (Myoseon Jang, UNC)
acid seed + decanol+ octanal non-acid seed + decanol+ octanal
To demonstrate the acid catalyzed aldehyde reaction, octanal was reacted directly on a ZnSe FTIR window by adding small amounts of aqueous H2SO4 acid catalyst solution (0.005 M). The spectra of the octanal/acid-catalyst system changed progressively as a function of time • The aldehydic C-H stretching at 2715 cm-1 immediately disappeared, the C=O stretching band at 1726 cm-1 gradually decreased • and the OH stretching at 3100-3600 cm-1 increased as hydrates formed.
Future research areas. • Determine the importance of particle phase reactions as a source of SOA. • Determine the importance of sesquiterpenes in SOA formation. • Clarification of the impact of drought and relative humidity on biogenic emissions is needed so these factors can be incorporated into emission models.