Dipole-Dipole Interactions
340 likes | 1.36k Views
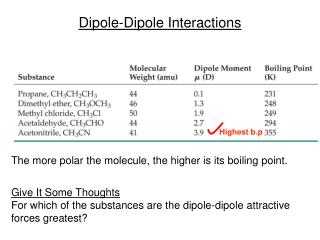
Dipole-Dipole Interactions. Highest b.p. The more polar the molecule, the higher is its boiling point. Give It Some Thoughts For which of the substances are the dipole-dipole attractive forces greatest?. Notes. Dipole-dipole forces are weaker than ion-dipole forces.

Dipole-Dipole Interactions
E N D
Presentation Transcript
Dipole-Dipole Interactions Highest b.p The more polar the molecule, the higher is its boiling point. Give It Some Thoughts For which of the substances are the dipole-dipole attractive forces greatest?
Notes • Dipole-dipole forces are weaker than ion-dipole forces • If two molecules have about the same mass and size, • then dipole-dipole forces increase with increasing polarity. • For molecules of similar polarity, • those with smaller volumes often have greater dipole-dipole attractions.
London Dispersion Forces While the electrons in the 1s orbital of helium would repel each other (and, therefore, tend to stay far away from each other), it does happen that they occasionally wind up on the same side of the atom.
London Dispersion Forces At that instant, then, the helium atom is polar, with an excess of electrons on the left side and a shortage on the right side.
London Dispersion Forces Another helium nearby, then, would have a dipole induced in it, as the electrons on the left side of helium atom 2 repel the electrons in the cloud on helium atom 1.
London Dispersion Forces London dispersion forces, or dispersion forces, are attractions between an instantaneous dipole and an induced dipole. • These forces are present in all molecules, whether they are polar or nonpolar. • The tendency of an electron cloud to distort in this way is called polarizability.
London Dispersion Forces • These forces are present in all molecules, whether they are polar or nonpolar. • The tendency of an electron cloud to distort in this way is called polarizability.
Larger surface area Factors Affecting London Forces Molecular shape • The shape of the molecule affects the strength of dispersion forces: long, skinny molecules (like n-pentane tend to have stronger dispersion forces than short, fat ones (like neopentane). • This is due to the increased surface area in n-pentane.
Factors Affecting London Forces Molecular Weight Size of the atom • The strength of dispersion forces tends to increase with increased molecular weight. • Larger atoms have larger electron clouds, which are easier to polarize.
a. increasing polarizability • CH4 < CI4 < CCl4 • CCl4 < CH4 < CI4 • CH4 < CCl4 < CI4 • CI4 < CCl4 < CH4 • List the substances CCl4, CI4, and CH4 in order of increasing • polarizability • Strength of dispersion forces
a. increasing polarizability • CH4 < CI4 < CCl4 • CCl4 < CH4 < CI4 • CH4 < CCl4 < CI4 • CI4 < CCl4 < CH4
Which Have a Greater Effect:Dipole-Dipole Interactions or Dispersion Forces? • If two molecules are of comparable size and shape, dipole-dipole interactions will likely be the dominating force. • If one molecule is much larger than another, dispersion forces will likely determine its physical properties.
Strong H-bonding in water How Do We Explain This? ? • The nonpolar series (SnH4 to CH4) follow the expected trend. • The polar series follows the trend from H2Te through H2S, but water is quite an anomaly. Boiling point as a function of molecular weight
Hydrogen Bonding A special case of dipole-dipole interaction
We call these interactions hydrogen bonds. Hydrogen Bonding • The dipole-dipole interactions experienced when H is bonded to N, O, or F are unusually strong. • Hydrogen bonding arises in part from the high electronegativity of nitrogen, oxygen, and fluorine. Examples of H bonding
Water How can we explain that ice floats on water Density of ice at 0 oC is 0.917 g/mL Density of water liquid at 0 oC is 1.00 g/mL Ice is less dense than water? H-Bonding
Hydrogen Bonding in water Ice hexagonal shape water dimer
The densities of liquid water and ice are identical. • The densities of liquid water and ice do not vary with temperature. • Ice is more dense than liquid water. • Ice is less dense than liquid water.
The densities of liquid water and ice are identical. • The densities of liquid water and ice do not vary with temperature. • Ice is more dense than liquid water. • Ice is less dense than liquid water.
Which of the following molecules can hydrogen bond with itself? • 1, 2 • 2, 3 • 3, 4 • 1, 2, 3 • 1, 2, 3, 4
Which of the following molecules can hydrogen bond with itself? • 1, 2 • 2, 3 • 3, 4 • 1, 2, 3 • 1, 2, 3, 4
Intermolecular and intramolecular hydrogen bonds play important role in our life • They are important in stabilizing protein structure, in DNA structure and function, etc. • An interesting consequence of H-bonding is that ice floats. Ice floats, so it forms an insulating layer on top of lakes, rivers, etc. Therefore, aquatic life can survive in winter. • Water expands when it freezes. • Frozen water in pipes may cause them to break in cold weather.