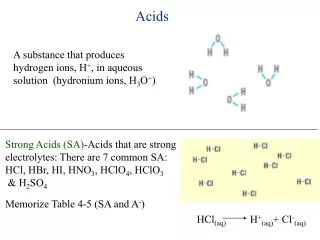
Understanding pH: The Measurement of Acidity and Basicity
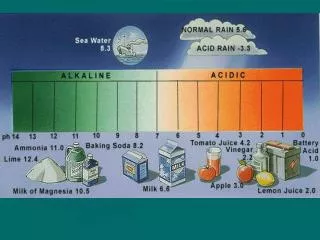
pH is a crucial measure that indicates how acidic or basic a solution is, ranging from 0 to 14. Solutions with pH values below 7 are considered acidic, while those above 7 are basic. A neutral solution has a pH of 7. The strongest acid has a pH close to 0, and the strongest base has a pH of 14. This activity involves creating a pH scale that illustrates various solutions, labeling acids, bases, and neutral solutions, and identifying strong versus weak substances. Engage with the pH scale activity for a hands-on understanding of acidity and alkalinity.

Understanding pH: The Measurement of Acidity and Basicity
E N D
Presentation Transcript
Background: • pH is a measure of how acidic or basic a solution is. The pH scale ranges from ______ to ______. Acidic solutions have pH values below _____. Basic or alkaline solutions have pH values above ____. Neutral solutions have a pH of ____. The strongest base would have a pH value of ______ and the strongest acid would have a pH of _______.
Copy the following on the back of page 43. pH Drawing instructions: • NEATLY Draw, Color, and Label your own pH scale. You must include: • Title (pH Scale) • (Drawings)1 Neutral solution; 5 bases; 5 Acids • Solutions must be drawn, colored, labeled and have its pH value written below it. • Label the Acids side, Base side and Neutral. • Label the strong verses weak acids and bases.
1.0 battery acid (sulfuric acid) 1.8-2.0 limes 2.2-2.4 lemon juice 2.2 vinegar (acetic acid) 2.8-3.4 fruit jellies 2.9-3.3 apple juice, cola 3.0-3.5 strawberries 3.7 orange juice 4.0-4.5 tomatoes 5.6 unpolluted rain 5.8-6.4 peas 6.0-6.5 corn 6.1-6.4 butter 6.4 cow's milk 6.5-7.5 human saliva 6.5-7.0 maple syrup 7.0 distilled water 7.3-7.5 human blood 7.6-8.0 egg whites 8.3 baking soda 9.2 borax 10.5 milk of magnesia 11.0 laundry ammonia 14 Drano pH Values of Household Items