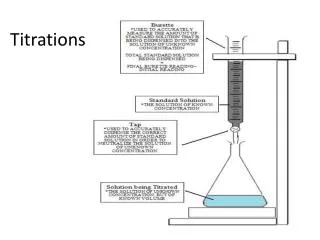
Redox Titrations
Redox Titrations. Oxidation-reduction reactions involve a transfer of electrons. The oxidising agent accepts electrons and the reducing agent gives electrons. In working out the equation for a redox reaction we can use the half-equation method.

Redox Titrations
E N D
Presentation Transcript
Oxidation-reduction reactions involve a transfer of electrons. The oxidising agent accepts electrons and the reducing agent gives electrons. In working out the equation for a redox reaction we can use the half-equation method. In this method we use the half-equation for the oxidising agent and the half-equation for the reducing agent then add them together.
Examples of half-reaction equations Iron(III) salts are reduced to iron(II) salts. The half equation is Fe3+ Fe2+ For the equation to balance the charge on the RHS must equal the charge on the LHS. This can be accomplished by inserting an electron on the LHS Fe3+ + e- Fe2+
When chlorine acts as an oxidising agent it is reduced to Cl- ions Cl2 2Cl- To obtain a balanced equation 2 electrons muct be inserted on the LHS Cl2 + 2e- 2Cl-
Potassium mangante(VII) is an oxidising agent. In acid solutions it is reduced to a manganese(II) salt MnO4- + 8H+ + 5e- Mn2+ + 4H2O Potassium dichromate(VI) is also an oxidising agent. It is reduced to a chromium(III) salt Cr2O7- + 14H+ + 6e- 2Cr3+ + 7H2O
Using half equations to obtain the equation for the reaction Find the equation for the reaction of iron(III) with chloride ions The 2 half reactions needed are 1 Fe3+ + e- Fe2+ 2 Cl2 + 2e- 2Cl- We need iron(III) and chloride as reactants so equation 2 must be reversed 3 2Cl- Cl2 + 2e-
Now we must balance the electrons, then add the half equations together to obtain the full equation. To balance the electrons we multiply equation 1 by 2 • Fe3+ + e- Fe2+ x2 4 2Fe3+ + 2e- 2Fe2+ 3 2Cl- Cl2 + 2e- 2Fe3+ + 2Cl- 2Fe2+ +Cl2 There must be the same no of electrons on each side which then cancel.
Construct an equation for the reaction of potassium manganate(VII) with ethanedioate ion Ethanedioate ion is C2O42- and the halfequation is C2O42- 2CO2 + 2e- 2MnO4- + 16H+ + 5C2O42- 2Mn2+ + 8H2O + 10CO2
Now we can do a titration calculation using this equation! A 25.0ml portion of sodium ethanedioate solution of concentration 0.200 mol/L is warmed and titrated against a solution of potassium manganate(VII). If 17.2ml of potassium manganate(VII) is required what is its concentration?
No of moles ethanedioate in 25.0ml of solution of conc 0.200 mol/L = 0.200 x 25 = 0.005000 mols 1000 Ratio of ethanedioate to manganate(VII) = 2:5 no of mols of manganate(VII) in 17.2ml = 0.005000 x 2 = 0.002000 mols 5 conc of manganate(VII) = 0.002000 x 1000 17.2 = 0.116 mol/L