WATER
WATER. WATER'S IMPORTANCE. Solvent Most molecules dissolved in water Reactant Water's involvement in hydrolysis reactions Product Water's involvement in condensation reactions Heat transfer medium E.g. boiling, steaming, cooling. WATER'S IMPORTANCE. Texture Juiciness, mouthfeel

WATER
E N D
Presentation Transcript
WATER'S IMPORTANCE • Solvent • Most molecules dissolved in water • Reactant • Water's involvement in hydrolysis reactions • Product • Water's involvement in condensation reactions • Heat transfer medium • E.g. boiling, steaming, cooling
WATER'S IMPORTANCE • Texture • Juiciness, mouthfeel • Snack foods • Vegetables • Meat • Preservation • Highly perishable foods usually have high water activity • E.g. bread vs. cracker or cereal • Economics • More water added = more $ UNDERSTANDING THE PHYSICAL AND CHEMICAL PROPERTIES OF WATER IS IMPORTANT IN THE STUDY OF FOOD AND PROCESSING
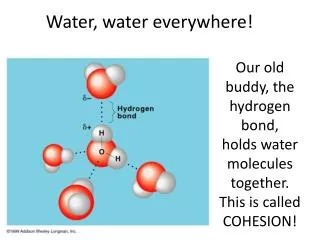
PHYSICAL & CHEMICAL PROPERTIES OF WATER • Water has very unique properties not shared by other similar hydrogen compounds or compounds of similar weight • Why? – this is explained by the unique structure of H2O
STRUCTURE OF WATER • Tetrahedral arrangement • Two free electrons of O act as H-bond acceptors while H acts as donor • Highly electronegative O pulls electrons from H, making H behave like a bare proton • Forms a dipole because of the electronegative O
Because of the DIPOLE and TETRAHEDRAL structure we can get strong H-bonding Water capable of bonding to 4 other water molecules Unique properties of water from other hydrides H-bond NOT a static phenomenon T dependent STRUCTURE OF WATER
WATER VAPOR • Water is “free” and devoid of any H-bonds • Large input of energy needed • an endothermic process • Large dissipation of same energy needed to make water lose kinetic energy • an exothermic process • Waters latent heat of vaporization is unusually high • to change 1 L from liquid to vapor need 539.4 kcal
LIQUID WATER • Extensively H-bonded • H-bond formation dependent on T • With increasing T get more mobility and increased fluidity
ICE • Forms when exactly 4 H-bonds are formed between water molecules • 2.78 A vs. 2.85 A in liquid • To get this order a lot of energy needs to be adsorbed by the environment • The strong H-bonding in ice forms an orderly hexagonal crystal lattice • 6 H2O molecules • Has 4X more thermal conductivity than water at same temperature
PROPERTIES OF ICE • Crystallization • Crystal growth occurs at freezing point • Rate of crystal growth decreases with decreasing temperature • Solutes slow ice crystal growth • Nucleation - affects ice crystal size. • Slow freezing results in few nucleation sites and large, coarse crystals • Fast freezing results in many nucleation sites and small, fine crystals • Heterogeneous nucleation • usually caused by a foreign particle, such as salt, protein, fat, etc. • Homogeneous nucleation • very rare, mainly occurs in pure systems
PROPERTIES OF ICE • SUPERCOOLING • Water can be cooled to temperatures below its freezing point without crystallization • When an ice crystal is added to supercooled water, temperature increases and ice formation occurs
Freezing induced changes in foods (examples) Destabilization of emulsions Flocculation of proteins Increased lipid oxidation Meat toughening Cellular damage Loss of water holding capacity PROPERTIES OF ICE Example: Effect of freezing on seafoods
WATER SOLUTE INTERACTIONS Association of water to hydrophilic substances • Bound water - occurs in vicinity of solutes • Water with highly reduced mobility • Water that usually won't freeze even at -40ºC • Water that is unavailable as a solvent • “Trapped” water • Water holding capacity • Hydrophilic substances are able to entrap large amounts of water • Jellies, jams, yogurt, jello, meat • Yogurt - often see loss of water holding as whey is released at the top of the yogurt
WATER SOLUTE INTERACTIONS • Ionic polar solutes • React readily with water and most are usually soluble in water • Water HYDRATES the ions • Charge interactions due to waters high DIELECTRIC CONSTANT • Can easily neutralize charges due to its high dipole moment • Large ions can break water structure • Have weak electric fields • Small ions can induce more structure in water • Have strong electric fields
WATER SOLUTE INTERACTIONS • Nonionic polar solutes • Weaker than water-ion bonds • Major factor here is H-bonding to the polar site • Example: SUCROSE • 4-6 H2O per sucrose • Concentration dependent • >30-40% sucrose all H2O is bound • T dependent solubility • C=O, OH, NH2 can also interact with each other and therefore water can compete with these groups • H-bond disrupters • urea - disrupts water Water bridge
WATER SOLUTE INTERACTIONS • Nonpolar • Unfavorable interaction with water • Water around non-polar substance is forced into an ordered state • Water affinity for water high compared to non-polar compound • Water forms a shell • Tries to minimize contact • Hydrophobic interactions • Caused because water interacts with other water molecules while hydrophobic groups interact with other hydrophobic groups
EFFECT OF SOLUTES ON WATER Boiling point • Vapor pressure is equal to atmospheric pressure • Strongly influenced by water - solute interaction • Solutes decrease vapor pressure and thus increase boiling point • Sucrose +0.52ºC/mol • NaCl +1.04ºC/mol ATMOSPHERIC PRESSURE VAPOR PRESSURE
EFFECT OF SOLUTES ON WATER 1 atm (sea level) mountains So does it take longer or shorter to boil an egg in the Rocky Mountains? Why?
EFFECT OF SOLUTES ON WATER Let's go back to our egg, what would happen if you added salt? Recommended that you add salt to water at high altitudes
EFFECT OF SOLUTES ON WATER Freezing point lowering • Freezing point can get extensive depression via solutes • Alter ability of water to form crystals due to H-bond disruption • Sucrose -1.86ºC/mol • NaCl -3.72ºC/mol • Eutectic pt - temp. • Where “all” water is frozen - usually around -50ºC • In most cases small amounts of water remains unfrozen (-20ºC) • These small patches of water can promote chemical reactions and damage
EFFECT OF SOLUTES ON WATER What explains all this? • Raoult's law P = P*/X1 or P*-P/P*= x/55.5M P = vapor pressure of solution; P* = vapor pressure pure solvent; X1 = mole fraction of solute; x = grams solutes in solution; 55.5M = moles of water per liter • This relationship is not only important for explaining the concepts of depressing freezing point and elevating boiling point • Also explains the concept of water activity
EFFECT OF SOLUTES ON WATER Osmotic pressure of solutions • There is a tendency for a system containing water and a solution separated with a membrane to be at equilibrium • The pressure needed to bring the two solutions at equilibrium is called OSMOTIC PRESSURE • The more the solution has of dissolved solutes (e.g. salt) the higher its osmotic pressure • Can use this in food processing and preparation • E.g. Crisping salad items • Increase turgor
Surface tension Water surface behaves differently than bulk phase Like an elastic film Due to unequal inward force Resist formation of a new surface thus forming surface tension EFFECT OF SOLUTES ON WATER
EFFECT OF SOLUTES ON WATER • Water has high surface tension • 72.75 dynes/cm (20ºC) • Because of the high surface tension special considerations are needed in food processing • To affect it one can: • Increase T (more energy) reduces surface tension • Add solutes • NaCl and sugars increase surface tension • Amphipathic molecules reduce surface tension
EFFECT OF SOLUTES ON WATER Ionization of water • Water can ionize into hydronium (H3O+) and hydroxyl (OH-) ions • Transfer of one proton to the unshared sp3 orbital of another water molecule • Pure water: Keq = Equilibrium (or ionization) constant Keq = [H3O]+ [OH]- [H2O] [H3O]+ [OH]- = Keq = Kw (Water dissociation constant) [10-7] [10-7] = [10-14]
EFFECT OF SOLUTES ON WATER • Acids and bases in food systems • Acid - proton donor • NH3 + H2O NH4+ + OH- • Base - proton acceptor • CH3COOH + H2O CH3COO- + H3O+ • Weak acids and bases • Most foods are weak acids • These constituents are responsible for buffering of food systems • Some examples • Acetic, citric, lactic, phosphoric, etc.
EFFECT OF SOLUTES ON WATER • Acids and bases in food systems • Is there a difference between weak and strong acids? • Strong acids • When placed in solution, 100% ionized • Weak acids • When placed in solutions weak acids form an equilibrium • HCl = H+ + Cl- • pH = -log [acid] = -log [H]+ HOAC H+ + OAC- pKa = -log Ka Keq = [H]+ [OAC]- [HOAC]
EFFECT OF SOLUTES ON WATER • Weak acids and bases • One cannot relate pH to concentration for weak acids and bases because of this equilibrium • One must understand how the acid behaves in solution • Knowing the dissociation constant of the acid is important to determine the effect on the pH of the system • The relationship of pH for weak acids and bases relies on the Henderson - Hasselback equation: pH = pKa + log [salt] [acid]
EFFECT OF SOLUTES ON WATER • Weak acids • Graphically behave like the figure when titrated with a strong base. The reverse holds true for weak bases What do we call this point?
EFFECT OF SOLUTES ON WATER • Buffering • Buffers resist changes in pH when acids and bases are added • Characteristics of a buffer • Maximum when pH = pKa or when [acid] = [salt] • Rule of thumb: pH = pKa ± 1 What is this point and its significance to food systems?
EFFECT OF SOLUTES ON WATER • Examples of natural pH control • Fruits - citric, malic, acetic, etc • Microbial control • Flavoring • Milk – pH around 6.5 • Controlled by three components • Phosphate, citrate, carbonate • Eggs • Fresh eggs - pH = 7.6 • After storage for several weeks - pH = 9-9.7 • Due to loss of CO2 • Problem - Loss of carbohydrate groups on proteins. Loss of protein functionality, causing decreased viscosity and poor foaming properties
EFFECT OF SOLUTES ON WATER • Examples of “man made” pH control • Food additives - ACIDULANTS • Citric acid - pectin jellies • pH must be around 2.9-3.0 • Also provides balance between tartness and sweetness • Yogurt and cottage cheese • Fermentation - glucose or lactose to lactic acid • pH reduction to around 4.6 will cause the gelation • Can add acidulants to imitate dairy yogurts - lactic, citric, phosphoric, HCl • Cheese • Alkaline salts of phosphoric acid to get good protein dispersion • Thermal process control • pH below 4.5 usually hinders C. botulinum growth • Less severe heat treatment required for these • Acidulants used to lower pH below 4.5 for some fruit and tomato products
EFFECT OF SOLUTES ON WATER • Examples of “man made” pH control • Acidulants - leavening agents • Used in the baking industry to give rise (release of CO2) - alternative to yeast • When HCO3- becomes acidic (pH < 6), CO2 forms, CO2 not very soluble so released as a gas Overall eq: H+ + HCO3-H2O + CO2
EFFECT OF SOLUTES ON WATER • Examples of “man made” pH control • Leavening systems • Bicarbonate (NaHCO3) - source of HCO3 and CO2 • Leavening acids • Drive bicarbonate (HCO3) to CO2 • Rate of acid release varies and therefore CO2 release • Phosphate - rapid release of CO2 • Sulfate – slow release of CO2 • Pyrophosphate - can be cleaved by phosphatases becoming more soluble - used in refrigerated doughs • d-Glucono-lactone - used in refrigerated doughs
EFFECT OF SOLUTES ON WATER • Examples of “man made” pH control • Acidulants - antimicrobials • pH is important for two reasons: 1. Solubility and 2. Activity • The salt is more soluble in aqueous systems • The acid is more active in its antimicrobial efficiency • Benzoic acid (0.05-0.1%) • Found naturally in prunes, cranberries, cinnamon and cloves • Active below pH 4 (active acidic form of the salt) • Highly soluble in the form of sodium salt • Effective - yeasts and bacteria, less for molds • Uses in acid foods - soft drinks, juices, pickles, dressings etc. • Parabens or r-hydroxybenzoate esters (0.05-0.1%) • Broader pH range (active at higher pH) • Mainly use methyl and propyl esters • Uses in baked goods, wines, pickles, jams, syrups, etc.
EFFECT OF SOLUTES ON WATER • Acidulants - antimicrobials • Sorbic acid (Na+ and K+ salt forms) (0.02-0.3%) • Max activity at pH 6.5; active at acid pH values • Most effective for yeast and molds • Inhibit, not inactivate • Uses in cheese, juices, wines, baked goods, etc. • Proprionic acid (proprionate) Ca2+ salt • Active up to pH 5 • Uses in breads (retards Bacillus) which causes ropiness in breads • Ropiness - thick yellow patches that can be formed into a rope-like structure making the bread inedible • Acetic acid • Nitrites and Nitrates • Sulfites
WATER ACTIVITY • What is meant by water activity? • Water has different levels of binding and thus activity or availability in a food sample • Simply put, Water activity (aw) helps to explain the relationship between perishability and moisture content • Greater moisture content faster spoilage (normally) • Why are there some perishable foods at the same moisture content that don't spoil at the same rate? • There is a correlation found between aw and various different spoilage and safety patterns
WATER ACTIVITY • Water has different levels of binding and thus activity or availability in a food sample • Food companies and regulatory agencies (e.g. FDA) rely on aw as an indicator of how fast and in what fashion a food product will deteriorate or become unsafe, and it also helps them set regulatory levels of aw for different foods Highly perishable foods aw > 0.9 Intermediate moist foods aw = 0.6-0.9 Shelf stable foods aw < 0.6
WATER ACTIVITY • Thermodynamic definition of aw • The tendency of water molecules to escape the food product from liquid to vapor defines the aw aw = p/pO=%RH/100 • Water activity is a measure of relative vapor pressure of water molecules in the head space above a food vs. vapor pressure above pure water • Scale is from 0 (no water) to 1 (pure water)
WATER ACTIVITY • Sorption isotherms • Help relate moisture content to aw • Each food has their own sorption isotherm • It is interesting that when water is added to a dry product, the adsorption is not identical to desorption • Some reasons • Metastable local domains • Diffusion barriers • Capillary phenomena • Time dependent equilibrium Temp. dependent
WATER ACTIVITY • Water sorption of a mixture • A mixture of two different food components with different aw leads to moisture migration from one food to another which can create problems • This is one reason why it is important to know the aw of a food product or ingredient • Examples: • Caramel, marshmallows and mints – all similar %moisture but very different aw • Fudge (aw = 0.65-0.75) covered with caramel (aw = 0.4-0.5) – what happens? • Granola bar with soft chewy matrix (aw = 0.6) and sugar coat (aw = 0.3)? • Hard candy (aw = 0.2-0.35) on a humid day?
WATER ACTIVITY • So, knowing the aw of a food component one can select the proper ingredients for a particular food product • For example, it is possible to create a multi-textured food product if components are added at the same aw
WATER ACTIVITY • Temperature dependency of the sorption isotherm can be a major problem and often overlooked Example: Crackers that experience a temperature rise during transportation At the same moisture content which would spoil faster?
WATER ACTIVITY • Sorption isotherms also explain the level of water binding in a food (i.e. types of water) • Type I: Tightly “bound” water (monolayer) • Unavailable/Unfreezable (at -40C) • Water - ion; water - dipole interactions • Type II: additional water layer (Vicinal water) • Slightly more mobility • Some solvent capacity • Type III: Water condensating in capillaries and pores (multilayer bulk-phase water) • More available (like dilute salt solution) • Can be entrapped in gels • Supports biological and chemical rections • Freezable True monolayer Monolayer
WATER ACTIVITY • Importance of aw in foods • Food stability directly related to aw • Influences storage, microbial growth, chemical & enzymatic deteriorations, etc. Vit C loss
WATER ACTIVITY aw is a major HURDLE for microorganisms but not the only one A) Microbial stability • Foods with aw > 0.9 require refrigeration because of bacteria spoilage • Exception: Very low pH Foods • Can control by making intermediate moisture foods (IMF) • Food with low aw to prevent microbial spoilage at room temp. But which can be eaten w/o hydration • Aw = 0.7 - 0.9 (20 -50% water) - achieved by drying or using solutes (sugar, salt) • dried fruits, jelly and jam, pet foods, fruity cakes, dry sausage, marshmallow, bread, country style hams • Minimal processing however preferred over IMF • Special problems • May need mold inhibitor • Lipid oxidation - may need antioxidant or inert packaging • Important in grains to prevent mold growth & possibly mycotoxin development • Must be below 0.8
WATER ACTIVITY B) Chemical stability • Maillard browning • Doesn't occur below type II water • Increases in type II water - water becomes a better solvent while reactants become more mobile • Reduced in type III - dilution or water is an inhibitor • Depends on food product (aw 0.53-0.55 in apple juice vs. 0.93 in anchovy) • Lipid oxidation • Low aw, lipid oxidation high - due to instability of hydroperoxides (HP) - unstable w/o water, no H-bonding • Slightly more addition of water stabilizes the HP and catalysts • Above type II water, water promotes the lipid oxidation rate because it helps to dissolve the catalysts for the reaction