Wacker Process
Jacob McNamara Elmhurst College, Elmhurst, Illinois. Problems with the old synthesis: -Originally produced for acetaldehyde -this was expensive due to the high temp. combustions of light hydrocarbons -28 cents per pound was the cost Solution to this problem:

Wacker Process
E N D
Presentation Transcript
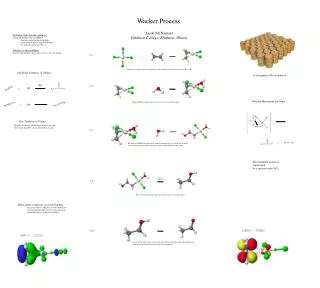
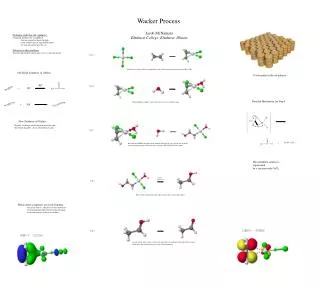
Jacob McNamara Elmhurst College, Elmhurst, Illinois Problems with the old synthesis: -Originally produced for acetaldehyde -this was expensive due to the high temp. combustions of light hydrocarbons -28 cents per pound was the cost Solution to this problem: -Started with ethylene, which only costs 3.5 cents per pound Wacker Process Step 1 + PdCl4 reacts with ethylene to form PdCl3 with a bond connecting to the bond of H2C=CH2 Old Initial Synthesis of Olefins Used mainly in the oil industry Step 2 + Detailed Mechanism for Step 4 This palladium complex reacts with water to eject a chlorine atom New Synthesis of Olefins: Replace a hydrogen on the ethylene molecule with the desired ligand by a basic substitution reaction. Step 3 + Based on the HOMO diagram below, another molecule of water attacks the pi-bond on the ethylene portion of the molecule, causing a CH2CH2OH chain to form. The palladium catalyst is regenerated by a reaction with CuCl2 Detailed Mechanism Step 4 Refer to the mechanism to the right for how this reaction takes place Metal-olefin complexes use back-bonding - this means there is a decrease in electron density of the pi-bonding orbital of the olefin and a gain of electron density in the pi-star orbital. Step 5 A molecule of water comes, and its lone pair takes the hydrogen atom off of the oxygen, which gives the final product, plus some hydronium ions.