Lens
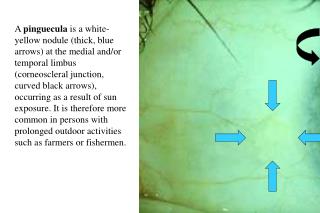
A pinguecula is a white-yellow nodule (thick, blue arrows) at the medial and/or temporal limbus (corneoscleral junction, curved black arrows), occurring as a result of sun exposure. It is therefore more common in persons with prolonged outdoor activities such as farmers or fishermen.

Lens
E N D
Presentation Transcript
A pinguecula is a white-yellow nodule (thick, blue arrows) at the medial and/or temporal limbus (corneoscleral junction, curved black arrows), occurring as a result of sun exposure. It is therefore more common in persons with prolonged outdoor activities such as farmers or fishermen.
A pterygium (arrows) is histologically identical to a pinguecula, but clinically is a triangular (“wing-shaped”) fibrovascular growth onto the corneal surface, which requires excision if it encroaches into the pupillary axis. It also is due to sun damage and typically occurs at the medial limbus of the eye.
Both pinguecula and pterygium occur as a result of solar damage to subepithelial collagen, characterized histologically by loss of eosinophilia, and fragmentation and clumping of these fibers. Although originally thought to be damage of elastic fibers, due to the positive reaction of fibers with a histochemical stain for elastin, pretreatment of the tissue with elastase fails to quench the positivity. Therefore, it is also called elastotic degeneration ("elastic-like") of stromal collagen. The overlying epithelium may be either atrophic, normal, or dysplastic.
Herpes simplex keratitis causes a characteristic dendritic (branching, arrows) ulcer of the corneal epithelium (here stained with rose bengal). Herpes simplex, once established, remains latent in the trigeminal ganglion and when triggered by stimuli such as cold or stress reactivates to cause recurrent bouts of ulceration. The cornea is hypesthetic (decreased sensation) due to the nerve involvement, and prone to superinfection by other organisms.
Recurrent bouts of herpes simplex keratitis may cause inflammation to extend into the corneal stroma (stromal keratitis), with subsequent vascularization and scarring. This is a histologic section of a corneal transplant from a patient with stromal keratitis. Note the vascularization (arrows) at all levels of the corneal stroma, with associated chronic inflammation.
Corneal dystrophies are inherited, bilateral, progressive, alterations of corneal structure that may involve any of the corneal layers. This is an example of Fuchs’ dystrophy of the corneal endothelium. Although there is no recognized familial inheritance pattern, it typically manifests as corneal edema and bullous keratopathy in elderly individuals, women more commonly than men. The clinical hallmark of Fuchs’ dystrophy is the development of central corneal guttata (“drops”), seen best on specular reflection of the endothelial surface, using the slit lamp (blue arrows).
Histologic section of a cornea with Fuchs’ dystrophy shows guttata to be flat-topped excrescences of Descemet’s membrane (PAS stain) with discontinuity of the endothelial mosaic allowing fluid into the corneal stroma.
Glaucoma is damage to the retinal nerve fibers associated with an increase in intraocular pressure. It can be divided into 2 major categories: angle-closure (or closed angle glaucoma) and open angle glaucoma. Aqueous humor, the principal determinant of intraocular pressure, is formed by the ciliary processes, flows from behind the iris through the pupil into the anterior chamber and then out through the trabecular meshwork or the suprachoroidal space. If the trabecular meshwork is physically blocked by the peripheral iris, the angle is considered closed. If it is not, then it is considered open.
Angle-closure glaucoma is an acute rise in pressure due to physical blockage of the anterior chamber angle structures, leading to symptoms of redness, ocular pain, photophobia, cloudy vision, and seeing haloes around lights. The angle blockage often occurs after the patient has been in a dark place for several hours (pupil dilated, peripheral iris apposed to trabecular meshwork).
This histologic section through the anterior portion of the eye demonstrates occlusion of the anterior chamber angle by the iris. Note the physical blockage of the trabecular meshwork by the peripheral iris. Cornea Anterior chamber Trabecular meshwork Iris Posterior chamber Lens Sclera Ciliary body
If not treated promptly, angle-closure glaucoma will also cause injury to retinal nerve fibers, but does not typically cause deep excavation of the optic disc. The affected disc, seen on the left, is pale (especially temporally, blue arrow) as compared to the normal disc on the right (black arrow), but is flat rather than excavated.
Primary open angle glaucoma is the most common type, occurring in about 4 % of the U.S. population over 40 years of age. It may escape detection until advanced injury has occurred, since it is insidious and often asymptomatic. The chronically increased intraocular pressure leads to loss of ganglion cells, the axons of which make up the nerve fiber layer and optic nerve. The optic disc becomes increasingly cupped, as shown in this fundus photograph (rim of cupping outlined by arrows).
A corresponding histologic section through the optic disc shows the excavation of the disc, with undermining of the edges (so-called "beanpot" cup).
Cataract is an opacity of the lens. This photograph shows one type of cataract, a posterior subcapsular cataract (arrows), often seen in patients on prolonged corticosteroid therapy. It may also be seen as an age-related phenomenon or in diabetics. Blue arrows indicate the posterior surface of the lens, yellow arrows the anterior surface, and white arrows outline the cataract.
Histologically, posterior subcapsular cataract is characterized by posterior migration and swelling of lens epithelial cells (so-called “bladder cells of Wedl”, blue arrows). Lens epithelium does not normally line the posterior lens capsule (black arrows) after birth.
Cataracts involving the lens cortex may assume a variety of shapes, but most commonly present as wedge-shaped white opacities (black arrows), with the apices of the wedge pointing toward the center of the lens.
Histologically, cortical cataracts are characterized by the fragmentation and rounding up of lens fibers, producing eosinophilic globules (“Morgagnian globules”). Since the lens fibers lose their nuclei during development, anuclear Morgagnian globules can be histologically differentiated from the nucleated bladder cells of Wedl.
Another type of age-related cataract is nuclear sclerosis. The central portion (nucleus) of the lens becomes yellow-brown (brunescent) and opaque. Nuclear sclerosis causes an increase in the refractive power of the eye; the elderly patient becomes more myopic and able to read again without bifocals or reading glasses (so-called "second sight"). Since the nuclear lens fibers are normally compacted with maturation, histologic differences between normal eyes and tbose with nuclear sclerotic cataracts are not readily recognizable. Yellow arrows mark the anterior lens surface, blue arrows the posterior lens surface, and white arrows the nuclear cataract.
Sympathetic ophthalmia is a granulomatous inflammation of the choroid in the fellow eye of a patient who has had a penetrating injury of one eye. The immune system reacts with an as yet undetermined antigen which was unmasked by the injury, and attacks the fellow ("sympathizing") uninjured eye. Enucleation of the injured eye within two weeks of injury will prevent the immune response. Fundoscopy (top) shows yellow-white subretinal nodules with a hazy view due to cells in the vitreous. Fluorescein angiography (bottom) demonstrates hyperfluorescence of the nodules due to increased vascular permeability in the areas of inflammation.
Histologic section of the choroid in sympathetic ophthalmia shows ill-defined aggregates of epithelioid histiocytes (i.e., granulomas, arrows) with surrounding lymphocytes.
The most common primary malignant eye neoplasm in adults is malignant melanoma of the uvea (uvea is the vascular layer of the eye and consists of the iris, ciliary body and choroid). Most melanomas occur in the posterior aspect of the eye (and therefore arise in the choroid), as seen in this fundus photograph. They typically have a dome-shaped or mushroom-shaped appearance, and pigmentation can be heavy, variable, or nonexistent.
Ultrasonography of melanomas will demonstrate their shape and relative density. On the left is a B-scan, showing a mass (white arrow) in the posterior aspect of the eye. On the right is an A-scan which shows the high peak (blue arrow) at the surface of the tumor, with low internal reflectivity (lower peaks following the initial spike).
MRI is also useful in documenting the shape and internal characteristics of uveal melanoma (hypointense to vitreous on T1-weighted image [yellow arrow]; hyperintense on T2-weighted image [blue arrow]), but most importantly may demonstrate extraocular extension (not seen here). T1 T2 Vitreous Vitreous
A gross photograph of a mushroom or collar-button shaped choroidal melanoma situated at the equator of the eye. When the tumor breaks through Bruch's membrane to expand into the subretinal space, the elastic nature of Bruch's membrane causes the focal constriction and mushroom shape of the tumor.
Along with size and extraocular extension, a major prognostic factor in melanoma is the histologic cell type. Uveal melanomas can be divided into spindle (good prognosis), epithelioid (poor prognosis) and mixed spindle-epithelioid (intermediate prognosis) categories. Spindle cells have elongate nuclei, longitudinal nuclear grooves and/or punctate nucleoli, and indistinct cell borders, as seen here
Epithelioid cells, in contrast to spindle cells, are polygonal cells with rounded nuclei, prominent central nucleoli, and distinct cell borders. Mitotic figures are usually more evident in epithelioid melanomas than in their spindle cell counterparts.
Melanomas gain access out of the eye principally through emissaria, channels carrying vessels and nerves that penetrate the sclera to supply the intraocular tissues. In this histologic section, melanoma cells are exiting the eye along an emissarial canal (arrows) containing an artery, the lumen of which is at the lower right. The vortex veins are the most common point of egress for choroidal melanomas. Metastasis of uveal melanomas is usually hematogenous, and the liver is the site most commonly involved.
The most common intraocular malignancy in adults is not primary, but secondary, i.e. metastasis. Lung and breast carcinomas are the most likely to spread to the eye. In contrast to primary uveal melanomas, metastases are often placoid (flat or shallowly elevated), as seen in this fundus photograph, and multiple. They typically are a near-terminal event; the average life expectancy after diagnosis of an intraocular metastasis is 6-9 months.
This histologic section demonstrates a choroidal metastasis of squamous cell carcinoma (lung primary). Commonly, metastatic lung cancer may present in the eye without the primary tumor being known, making the clinical distinction between primary and secondary tumors in the eye difficult. Any patient presenting with an intraocular mass should have a systemic workup to exclude metastasis from another site.
Cytomegalovirus retinitis typically occurs in immunocompromised patients, particularly AIDS. The infection usually tracks along retinal vasculature (left), causing opacification and hemorrhage; variant clinical manifestations may include macular disease (right).
Histologic section of a retina infected with CMV. Note the large cells with prominent basophilic "owl's eye" intranuclear inclusion (arrows).
Background diabetic retinopathy is characterized by numerous microaneurysms, dot hemorrhages and hard exudates, and occasional cotton-wool spots (blue arrow) in the posterior fundus. Most of the red dots seen in this picture represent hemorrhages; microaneurysms are more easily seen on fluorescein angiography.
Fluorescein angiography (same field as the previous fundus photograph) is much more sensitive in identifying microaneurysms and is the method of choice for following patients. It will also highlight areas of capillary nonperfusion and any neovascularization.
Trypsin digested (to remove nonvascular tissue) ink injected whole mount preparations of retina highlight microaneurysms (blue arrows) of the capillary bed and areas of nonperfusion (yellow arrows). The initial pathologic alteration is loss of pericytes in the capilary walls, followed by loss of endothelial cells and lack of blood flow.
Preproliferative diabetic retinopathy is heralded by venous beading and tortuosity, numerous cotton-wool spots (arrows), and intraretinal microvascular abnormalities ("IRMA's"). Patients who develop these changes are at high risk for subsequent neovascularization, and should be treated with panretinal photocoagulation.
Formations of new vessels (neovascularization, "NV") at the optic disc ("NVD") or elsewhere in the retina ("NVE") are the hallmark of proliferative diabetic retinopathy. The new vessels may grow on the surface of the retina, but often grow into the vitreous. These vessels are abnormal and bleed easily, leading to reparative fibrovascular tissue ingrowth, contraction, and tractional retinal detachment. NVD
Photomicrograph of neovascularization along the surface of the retina (black arrow). Note the wrinkling of the retina, particularly the photoreceptors (white arrows), resulting in focal tractional retinal detachments.
Proliferative diabetic retinopathy with early (left) and established (right) fibrovascular tissue ingrowth (so-called “retinitis proliferans”).
A gross photograph (left) of a complete funnel-shaped retinal detachment extending forward from the optic disc in an enucleated eye. A higher magnification (right) of an in vivo fibrovascular tissue membrane (arrows) into the vitreous in a patient with proliferative diabetic retinopathy.
A low power histologic section demonstrates a closed funnel-shaped complete retinal detachment. The subretinal space (blue arrows) is filled with serosanguinous fluid. Contracture of the fibrovascular tissue has pulled the retina forward to occupy the retrolental space (black arrows). The choroid and ciliary body are artifactually separated from the sclera (yellow double arrows).
Panretinal laser photocoagulation inhibits neovascularization by obliterating the ischemic retina and circumventing production of angiogenic factors. The fundus photo on the left shows the results of laser burns in a grid pattern shortly after treatment; at a later date the retinal pigment epithelium has hypertrophied and caused the spots to become pigmented.
A histologic section through a photocoagulation burn shows focal loss of photoreceptors [PR] and outer nuclear layer [ONL], with chorioretinal adhesion (curved blue arrows)and retinal pigment epithelial hypertrophy (straight blue arrows). ONL ONL PR PR
Even in advanced proliferative diabetic retinopathy (left) (“retinitis proliferans”), vitrectomy (surgical removal of vitreous and fibrovascular tissue) may allow the retina to become reattached and recover some degree of vision (right).
The fundus picture in accelerated ("malignant") hypertension is similar to that in central retinal vein occlusion, although it is bilateral and predominantly localized to the posterior fundus. In addition, hard exudates (small yellow dots) are present in the macula.
Histologic sections of cotton-wool spots show microinfarcts with swelling of nerve fibers ("cytoid bodies") in the nerve fiber layer (left). The swelling results from axoplasmic stasis and accumulation of intracellular organelles such as mitochondria. On the right the cytoid bodies (arrows) are highlighted by ink injected into the ganglion cells.
Histologic section of a hard exudate (blue arrow) shows its characteristic location in the outer plexiform layer of the retina. It is the result of leakage of lipids and proteins into the retina without complete resorption.[ONL=outer nuclear layer; OPL=outer plexiform layer; INL=inner nuclear layer] INL OPL ONL
Fundus appearance in central retinal artery occlusion shows a cherry red spot in the fovea (arrow) due to edema (without hemorrhage) of the surrounding retina. The fovea, being the thinnest area of the retina, allows the red reflex of the choroid to show through. Central retinal artery occlusion causes immediate, complete blindness and is irreversible if untreated within hours.
Histologic cross section of an optic nerve demonstrates a thrombo-embolus occluding the central retinal artery (blue arrow), and recanalization of the central retinal vein (black arrow) indicating prior occlusion of that vessel. Vein occlusions most commonly are due to compression by a diseased artery (hypertension, atherosclerosis), since the two share a common adventitial sheath. This histologic section, therefore, shows features indicating both central retinal artery and vein occlusion.
A fundus photograph of central retinal vein occlusion shows marked hemorrhage and cotton-wool spots throughout the retina (so-called "strawberry sundae”appearance), with edema of the optic nerve head.
A histologic section in vein occlusion shows intraretinal hemorrhages in nearly all layers of the retina and in the subretinal space, resulting in a hemorrhagic retinal detachment. Retina Subretinal space Choroid