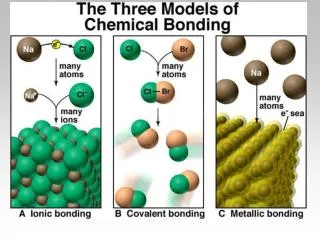
Figure 9.11: Potential-energy curve for H 2 .
Figure 9.11: Potential-energy curve for H 2 . Covalent Bonding in Hydrogen, H 2. For animation, open, then click on "reaction of sodium with chlorine“ Then choose “magnify the reaction. Figure 9.10: The electron probability distribution for the H 2 molecule.

Figure 9.11: Potential-energy curve for H 2 .
E N D
Presentation Transcript
For animation, open, then click on "reaction of sodium with chlorine“ Then choose “magnify the reaction.
Figure 9.10: The electron probability distribution for the H2 molecule. Animation and choose covalent bonding between hydrogens
Covalent bonds http://wine1.sb.fsu.edu/chm1045/notes/Bonding/Covalent/Bond04.htm animation http://www.chem.ox.ac.uk/vrchemistry/electronsandbonds/intro1.htm
For elements larger than Boron, atoms usually react to develop octets by sharing electrons. H, Li and Be strive to “look” like He. B is an exception to the noble gas paradigm. It’s happy surrounded by 6 electrons so the compound BH3 is stable. Try drawing a Lewis structure for methane.
Draw Lewis dot structures for the halogens. Notice that these all follow the octet rule! Try oxygen and nitrogen. These also follow the octet rule!
Figure 9.12: Molecular model of nitroglycerin. What is the formula for this compound?
Rules for drawing Lewis structures 1. Count up all the valence electrons 2. Arrange the atoms in a skeleton 3. Have all atoms develop octets (except those around He)
Make some Lewis Dot Structures with other elements: SiH4 H2O NH3 CH2O C2H6 C2H6O
Make some Lewis Dot Structures with other elements: CH4 H2O NH3 CH2O C2H6 C2H6O
Rules for drawing Lewis structures 1. Count up all the valence electrons 2. Arrange the atoms in a skeleton 3. Have all atoms develop octets (except those around He) 4. Satisfy bonding preferences!
Draw a Lewis structure for C2H4 Draw a Lewis structure for C2H2 Try COCl2
The Relation of Bond Order,Bond Length and Bond Energy Bond Bond Order Average Bond Average Bond Length (pm) Energy (kJ/mol) C O 1 143 358 C O 2 123 745 C O 3 113 1070 C C 1 154 347 C C 2 134 614 C C 3 121 839 N N 1 146 160 N N 2 122 418 N N 3 110 945 Table 9.4
Determining Bond Polarity from Electronegativity Values Problem: (a)Indicate the polarity of the following bonds with a polarity arrow: O - H, O - Cl, C - N, P - N, N - S, C - Br, As - S (b) rank those bonds in order of increasing polarity. Plan: (a) We use Fig. 9.16 to find the EN values, and point the arrow toward the negative end. (b) Use the EN values. Solution: a) the EN of O = 3.5 and of H = 2.1: O - H the EN of O = 3.5 and of Cl = 3.0: O - Cl the EN of C = 2.5 and of P = 2.1: C - P the EN of P = 2.1 and of N = 3.0: P - N the EN of N = 3.0 and of S = 2.1: N - S the EN of C = 2.5 and of Br = 2.8: C - Br the EN of As = 2.0 and of O = 3.5: As - O b) C - Br < C - P < O - Cl < P - N < N - S < O - H < As - O 0.3 < 0.4 < 0.5 < 0.9 < 0.9 < 1.4 < 1.5
Percent Ionic Character as a Function ofElectronegativity Difference (En) Fig. 9.19
Lewis Structures of Simple Molecules H H C H H .. .. F .. .. .. F C F .. .. .. .. F H H .. CH4 .. H C C O H Methane H H Ethyl Alcohol (Ethanol) .. .. .. .. O K+ .. Cl .. .. .. .. .. .. O O .. CF4 .. KClO3 Potassium Chlorate Carbon Tetrafluoride
Resonance: Delocalized Electron-Pair Bonding - I Ozone : O3 .. .. .. .. .. O O .. .. .. .. .. .. O O O O .. II I Resonance Hybrid Structure .. O .. .. .. .. O O One pair of electron’s resonances between the two locations!!
Resonance: Delocalized Electron-Pair Bonding - II H H C C H H C C C C H H C C H H C C H H H C C C H H C H H C H C C H C Benzene Resonance Structure H
Lewis Structures of Simple Molecules Resonance Structures -III Nitrate .. .. .. O N .. .. .. .. .. O O .. .. .. O .. .. O N N .. .. .. .. .. .. .. O O .. .. .. .. O O
.. .. Cl .. .. B .. .. .. .. Cl Cl Lewis Structures for Octet Rule Exceptions .. .. .. .. .. .. F .. .. .. .. F Cl .. .. F Each chlorine atom has 8 electrons associated. Boron has only 6! Each fluorine atom has 8 electrons associated. Chlorine has 10 electrons! . .. .. .. .. .. .. N .. .. .. .. Cl Be Cl .. O O Each chlorine atom has 8 electrons associated. The beryllium has only 4 electrons. NO2 is an odd electron atom. The nitrogen has 7 electrons.
Resonance Structures - Expanded Valence Shells .. .. O O .. .. .. .. H O S O H H O S O H .. .. O O .. .. .. .. .. .. .. .. .. .. .. .. .. .. F F F F .. .. .. .. .. .. .. F S F P .. .. F .. F .. .. .. .. .. .. .. .. F F .. F p = 10e- S = 12e- Sulfur hexafluoride Phosphorous pentafluoride .. .. .. .. .. Resonance Structures .. .. .. .. .. Sulfuric acid S = 12e-
Lewis Structures of Simple Molecules . . Sulfate . . -2 O . . . . Resonance Structures-V . . . . O S O . . . . -2 . . . . O o * o o o o O Plus 4 others for a total of 6 x o o o o o x O S O o * x o o o . . x o o -2 o o . . . . O x x . . . . o o O o o O S O o o . . . . . . . . O . . x = Sulfur electrons o = Oxygen electrons
VSEPR: Valence Shell Electron Pair Repulsion: A way to predict the shapes of molecules Pairs of valence electrons want to get as far away from each other as possible in 3-dimensional space.
Balloon Analogy for the MutualRepulsion of Electron Groups Two Three Four Five Six Number of Electron Groups
AX2 Geometry - Linear .. .. .. .. Molecular Geometry = Linear Arrangement .. .. Cl Be Cl BeCl2 1800 Gaseous beryllium chloride is an example of a molecule in which the central atom - Be does not have an octet of electrons, and is electron deficient. Other alkaline earth elements also have the same valence electron configuration, and the same geometry for molecules of this type. Therefore this geometry is common to group II elements. .. .. .. .. O C O CO2 1800 Carbon dioxide also has the same geometry, and is a linear molecule, but in this case, the bonds between the carbon and oxygens are double bonds.
The Two Molecular Shapes of the Trigonal Planar Electron-Group Arrangement
S O O AX3 Geometry - Trigonal Planar .. .. .. .. All of the boron Family(IIIA) elements have the same geometry. Trigonal Planar ! .. .. F F BF3 B Boron Trifluoride 1200 .. .. .. F AX2E SO2 .. - .. .. .. .. O .. .. NO3- 1200 .. .. N .. .. .. .. O O The AX2E molecules have a pair of Electrons where the third atom would appear in the space around the central atom, in the trigonal planar geometry. 1200 Nitrate Anion
The Three Molecular Shapes of the Tetrahedral Electron-Group Arrangement
107.30 .. N N H H H AX4 Geometry - Tetrahedral H Methane H 109.50 CH4 H C H C H H H H All molecules or ions with four electron groups around a central atom adopt the tetrahedral arrangement H H 109.50 109.50 + H+ H Ammonia is in a tetrahedral shape, but it has only an electron pair in one location, so the smaller angle! all angles are the same! Ammonium Ion H
The Four Molecular Shapes of the Trigonal Bipyramidal Electron- Group Arrangement