Molecular Geometry (p. 232 – 236)
Ch. 8 – Molecular Structure. Molecular Geometry (p. 232 – 236). A. VSEPR Theory. V alence S hell E lectron P air R epulsion T heory Electron pairs orient themselves in order to minimize repulsive forces. Lone pairs repel more strongly than bonding pairs!!!. A. VSEPR Theory.

Molecular Geometry (p. 232 – 236)
E N D
Presentation Transcript
Ch. 8 – Molecular Structure Molecular Geometry(p. 232 – 236)
A. VSEPR Theory • Valence Shell Electron Pair Repulsion Theory • Electron pairs orient themselves in order to minimize repulsive forces
Lone pairs repel more strongly than bonding pairs!!! A. VSEPR Theory • Types of e- Pairs • Bonding pairs – form bonds • Lone pairs – nonbonding e- • Total e- pairs– bonding + lone pairs
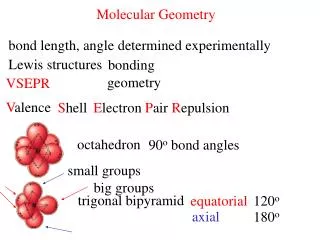
Bond Angle Bond Angle A. VSEPR Theory • Lone pairs reduce the bond angle between atoms
Know the 13 common shapes & their bond angles! B. Determining Molecular Shape • Draw the Lewis Diagram • Tally up e- pairs on central atom (bonds + lone pairs) • double/triple bonds = ONE pair • Shape is determined by the # of bonding pairs and lone pairs
BeH2 C. Common Molecular Shapes 2 total 2 bond 0 lone → Electronic Geometry = linear Hybridization = sp LINEAR 180°
BF3 C. Common Molecular Shapes → Electronic Geometry = trigonal planar Hybridization = sp2 3 total 3 bond 0 lone TRIGONAL PLANAR 120°
NO21- C. Common Molecular Shapes → Electronic Geometry = trigonal planar Hybridization = sp2 3 total 2 bond 1 lone BENT <120°
CH4 C. Common Molecular Shapes → Electronic Geometry = tetrahedral Hybridization = sp3 4 total 4 bond 0 lone TETRAHEDRAL 109.5°
NCl3 C. Common Molecular Shapes → Electronic Geometry = tetrahedral Hybridization = sp3 4 total 3 bond 1 lone TRIGONAL PYRAMIDAL 107° <109.5°
H2O C. Common Molecular Shapes → Electronic Geometry = tetrahedral Hybridization = sp3 4 total 2 bond 2 lone BENT 104.5° <109.5°
PI5 C. Common Molecular Shapes → Electronic Geometry = trigonal bipyramidal Hybridization = dsp3 5 total 5 bond 0 lone TRIGONAL BIPYRAMIDAL 120°/90°
KrF4 C. Common Molecular Shapes → Electronic Geometry = trigonal bipyramidal Hybridization = dsp3 5 total 4 bond 1 lone SEESAW <120°/<90°
ClF3 C. Common Molecular Shapes → Electronic Geometry = trigonal bipyramidal Hybridization = dsp3 5 total 3 bond 2 lone T-SHAPE <90°
I31- C. Common Molecular Shapes → Electronic Geometry = trigonal bipyramidal Hybridization = dsp3 5 total 3 bond 2 lone LINEAR 180°
SH6 C. Common Molecular Shapes → Electronic Geometry = octahedral Hybridization = d2sp3 6 total 6 bond 0 lone OCTAHEDRAL 90°
IF5 C. Common Molecular Shapes → Electronic Geometry = octahedral Hybridization = d2sp3 6 total 5 bond 1 lone SQUARE PYRAMIDAL <90°
SF4 C. Common Molecular Shapes → Electronic Geometry = octahedral Hybridization = d2sp3 6 total 4 bond 2 lone SQUARE PLANAR 90°
O O Se O D. Examples • SeO3 3 total 3 bond 0 lone E.G. = TRIGONAL PLANAR M.G. = TRIGONAL PLANAR 120°
H As H H D. Examples • AsH3 4 total 3 bond 1 lone E.G. = TETRAHEDRAL M.G. = TRIGONAL PYRAMIDAL 107° (<109.5°)
E. Hybridization • Provides information about molecular bonding and molecular shape • Several atomic orbitals mix to form same total of equivalent hybrid orbitals
E. Hybridization • Carbon is common example (orbital diagram) • One of 2s electrons is promoted to 2p • 4 identical orbitals form sp3 hybridization
Remember the subscript is the orbital, not e- configuration! E. Hybridization • Other types of hybridization • Be – 2 ve- forms sp • Al – 3 ve- forms sp2 • Si – 4 ve- forms sp3 • Kr – 8 ve- forms dsp3 • S – 6 ve- forms d2sp3 exceptions
F. Hybridization Example • Compare shapes and hybrid orbitals: PF3 PF5