THE TRANSITION METALS
THE TRANSITION METALS. Transition means “an in between state” and the transition elements come in between Group 2 and Group 3. Gp 2. Gp 3. H. He. Li. Be. B. C. N. O. F. Ne. Na. Mg. Al. Si. P. S. Cl. Ar. K. Ca. Sc. Ti. V. Cr. Mn. Fe. Co. Ni. Cu. Zn. Ga. Ge.

THE TRANSITION METALS
E N D
Presentation Transcript
THE TRANSITION METALS
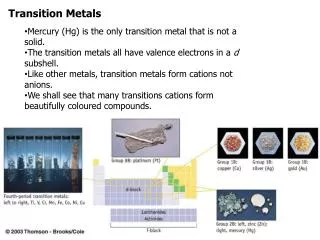
Transition means “an in between state” and the transition elements come in between Group 2 and Group 3. Gp 2 Gp 3 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt ? ? ? The Transition elements Transition Elements
They often act as catalysts. They have high melting points. They have high density. They are less reactive than Group 1 or Group 2 metals. They mostly form coloured compounds. Transition metals are often referred to as ‘typical’ metals. General Characteristics Transition Elements
Similarities are more noticeable than differences although there are still some broad patterns. They are all dense which is what we expect of metals. Sc Ti V Cr Mn Fe Co Ni Cu Zn Properties – density
Melting points show no regular pattern – other than nearly all being high which is typical of metals. (Note zinc doesn’t fit very well on either density or melting point.) Sc Ti V Cr Mn Fe Co Ni Cu Zn Properties – melting point
Again it is similarities that stand out rather than differences: they tend to react relatively slowly (e.g.) with air, water and acid. The general trend is to reduced activity across the Periodic Table but again the trend is far from perfect with zinc in particular being more reactive than you might expect. Sc Ti V Cr Mn Fe Co Ni Cu Zn Properties – reactivity Rusting: a slow but costly process! General reduced reactivity
A catalyst is a substance that speeds up a chemical reaction without being used up. Catalysts are hugely valuable in industry where they can save time and energy. Many transition elements ( and their compounds) are catalysts. Ni Ti V Fe Used in plastic manufacture Used in oil hydrogenation Properties – catalysis
The three most commonly known transition elements are iron or steel, copper and zinc. iron or steel copper zinc Electrical and plumbing work General engineering metal Galvanising steel to protect it Uses
Pair the metal up with its uses copper iron or steel zinc
Pair the metal catalyst with the substance. Fe Ni Ti V
Pair the statement about the transition elements with the words.
Which is a true statement about most transition elements? They are non-metals. They are light (low density). They are strong. They are non-conductors.
Which of these does copper NOT tend to be used for? Plumbing work. Electrical work Ornaments and jewellery Tools
Transition elements can speed up other reactions without getting used up in the process. What do we call substances that do this? Capitalists Catalysts Catholics Catapults
Transition elements often form coloured compounds. What colour compound does copper usually form? Blue Yellow Red Violet
How would you describe the reactivity of transition elements within the Periodic Table? Always more reactive across periods (left to right). Generally less reactive across periods (left to right). No change in reactivity. Changes but no trends in reactivity.