Plasmids and Vectors
Plasmids and Vectors. Instructor Supplement to pGlo Bacterial Transformation. Promotor Site. Origin of Replication. Antibiotic Resistance Gene. Multiple Cloning Site. A more detailed look at plasmids. Cloning into a Plasmid. Asilomar Conference.

Plasmids and Vectors
E N D
Presentation Transcript
Plasmids and Vectors Instructor Supplement to pGlo Bacterial Transformation
Promotor Site Origin of Replication Antibiotic Resistance Gene Multiple Cloning Site A more detailed look at plasmids
Asilomar Conference • People believed that “safe” strains of bacteria, viruses and vectors could be made in a few weeks • NIH formed the Recombinant DNA Advisory Committee (RAC) • It took 1 year (1976) before the first “safe” (EK2 category) line of E. coli was released • That year, RAC released a set of guidelines requiring the use of safe bacteria
NIH Guidelines • Self Regulation in Science Milestone • Contents • Specified handling and construction processes • Microorganisms containing recombinant DNA were prohibited outside of the laboratory • Vectors that sexually move to “unsafe” bacteria was prohibited • Subsequent modifications • 1986 expanded to include animals and plants, and 4 biosafety levels • 1994 officially relinquished control of GMO plants in the environment to EPA and APHIS
The First “Safe” Bacterium • Released in 1976 by Roy Curtiss III at the University of Alabama • E. coli 1776 • Required diaminopimelic acid (DAP) • Fragile cell walls (low salt, detergent sensitive) • Difficult to work with • Slow grower • Poor receptor for transformation
In the 1970’s and 1980’s • The first cloning vectors such as pSC101 had limited functionality • The next trend was to develop smaller plasmids • Advantages • Increased efficiency of transformation • Easier to restriction map • Higher copy numbers
The Cadillac of Cloning Vectors • pBR322 • Clone fragment in one antibiotic gene • Select for other antibiotic resistance • Screen for presence of one resistance gene (selects against untransformed bacteria) and loss of resistance to interrupted antibiotic resistance gene (selects for recombinant molecule) EcoRI TetR AmpR pBR322 4,361 bp APstI BamHI
Next Major Advance in Plasmid(ology) • The inclusion of polylinkers into plasmid vectors • Polylinker is a tandem array of restriction endonuclease sites in a very short expanse of DNA • For example, pUC18’s polylinker • Sites for 13 RE’s • Region spans the equivalent of 20 amino acids or 60 nucleotides Source: Bio-Rad Laboratories
The Polylinker Advantage • Unique sites (usually) • Insert excision facilitated • Restriction endonuclease mapping and Subcloning made easier
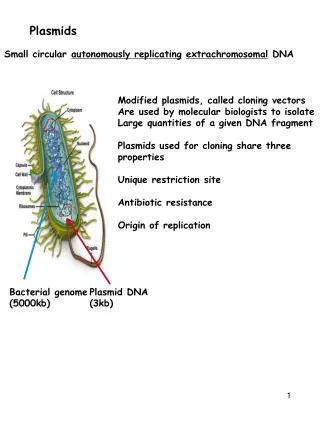
Features of many modern Plasmids • Small size • Origin of replication • Multiple cloning site (MCS) • Selectable marker genes • Some are expression vectors and have sequences that allow RNA polymerase to transcribe genes • DNA sequencing primers
The Major Limitation of Cloning in Plasmids • Upper limit for clone DNA size is 12 kb • Requires the preparation of “competent” host cells • Inefficient for generating genomic libraries as overlapping regions needed to place in proper sequence • Preference for smaller clones to be transformed • If it is an expression vector there are often limitations regarding eukaryotic protein expression
Bacteriophage lambda (λ) • A virus that infects bacteria • In 1971 Alan Campbell showed that the central third of the genome was not required for lytic growth. People started to replace it with E. coli DNA
Lambda genome is approximately 49 kb in length. Only 30 kb is required for lytic growth. Thus, one could clone 19 kb of “foreign” DNA. Packaging efficiency 78%-100% of the lambda genome. A complete animation of the lytic cycle: http://www.blackwellpublishing.com/trun/artwork/Animations/Lambda/lambda.html
Bacteriophage lambda • Protein capsule of lambda has a tight constraint on the amount of DNA that will fit inside it (~ 55kb) • By the early 1970’s we knew that a good portion of lambda was not required • “Junk” DNA COS site: Cohesive “sticky” ends Lysis Head Replication ori Tail Circularized lambda Lysogeny
COS Lysis Head Replication ori Tail Not Quite Bacteriophage lambda • Eliminate the non-essential parts of lambda • Can now insert large pieces of DNA (~ 20 kb)
Lambda was great: • Larger insert size • Introducing phage DNA into E.coli by phage infection is much more efficient than transforming E.coli with plasmid DNA But: • Have to work with plaques
ori 21.5 kb TetR cos Cos site is the only requirement for packaging into phage particle EcoRI Cosmids • Hybrid vectors: plasmids that contain bacteriophage lambda cos sites • DNA (~ 33-48 kb) cloned into restriction site, the cosmid packaged into viral particles and these phages used to infect E.coli • Cosmid can replicate in bacterial cell, so infected cells grow into normal colonies • Insert DNA limited by the amount of DNA that can fit into phage capsule • Somewhat unstable, difficult to maintain
Other Vectors • BACs (Bacterial artificial chromosomes) • Large low copy number plasmids (have ori and selectable marker) • Can be electroporated into E. coli • Useful for sequencing genomes, because insert size 100 - 300kb • YAC (Yeast Artificial Chromosome) • Can be grown in E.coli and Yeast • Miniature chromosome (contains ori, selectable markers, two telomeres, and a centromere • Can accept 200 kb -1000 kb; useful for sequencing • Ti plasmids; to introduce genes into plants • Expression vectors
How do you identify and clone a gene of interest? • Screen A DNA library: • Genomic • cDNA • Use Polymerase Chain Reaction (PCR) to clone gene of interest
What can you do with a library? • Can be used to complement a mutant (this is more common for research in bacteria). • Can use it in a colony hybridization.
Screening libraries by colony hybridization