Mass spectrometry 8/23/12
Mass spectrometry 8/23/12. What are the principles behind MS? What do all MS instruments have in common? What are the different types of MS?. Lecture outline: Introduction to mass spectrometry sample introduction systems, mass analyzers popular combinations in geosciences.

Mass spectrometry 8/23/12
E N D
Presentation Transcript
Mass spectrometry 8/23/12 What are the principles behind MS? What do all MS instruments have in common? What are the different types of MS? • Lecture outline: • Introduction to mass spectrometry • sample introduction systems, mass • analyzers • popular combinations in geosciences JJ Thomson’s cathode ray tube, 1897
Introduction to Mass Spectrometry Sample introduction Count ions Separate masses Collect results Ionization Minimize collisions, interferences Nier-type mass spec
Basic equations of mass spectrometry Ion’s kinetic E function of accelerating voltage (V) and charge (z). Centrifugal force Applied magnetic field balance as ion goes through flight tube Combine equations to obtain: Fundamental equation of mass spectrometry Change ‘mass-to-charge’ (m/z) ratio by changing V or changing B. NOTE: if B, V, z constant, then:
If: B in gauss r in centimeters m in amu V in volts z in electronic charge then…. What magnetic field strength would be required to focus a beam of CO2+ ions on a collector of a mass spectrometer whose analyzer tube as a radius of 31.45cm, assuming a voltage of 1000V? Change your magnetic field strength by -10%, what voltage puts the CO2 ions into the collector?
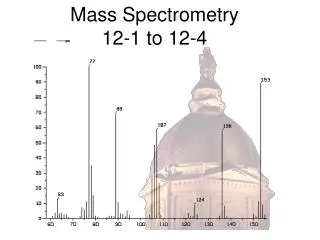
Examples of mass spec data output Ex: B You can scan in B or V to sweep masses across a single detector. OR You can put different masses into multiple cups without changing B or V.
Sample Introduction Systems (aka “front ends”) 1) Gas source (lighter elements) dual inlet - sample purified and measured with standard gas at identical conditions precisions ~ ±0.005% continous flow - sample volatized and purified (by EA or GC) and injected into mass spec in He carrier gas, standards measured before and after, precisions ~ 0.005-0.01% 2) Solid source (heavier elements) TIMS - sample loaded onto Re filament, heated to ~1500°C, precisions ~0.001% laser ablation - sample surface sealed under vacuum, then sputtered with laser precisions ~0.01%? 3) Inductively coupled plasma (all elements, Li to U) ICPMS - sample converted to liquid form, converted to fine aerosol in nebulizer, injected into ~5000K plasma torch
Ionization occurs in the ‘source’ Electron Ionization Gas stream passes through beam of e-, positive ions generated. Thermal Ionization Plasma: Gas stream passes through plasma maintained by RF current and Ar. Themal: Filament heated to ~1500C
Mass Analyzers - the quadrupole vs. magnetic sector Quadrupole: Changes DC and RF voltages to isolate a given m/z ion. PRO: cheap, fast, easy Magnetic Sector: Changes B and V to focus a given m/z into detector. PRO: turn in geometry means less ‘dark noise’, higher precision
Two types of ion detectors A) Faraday collector - long life, stable, for signals > 2-3e6 cps B) Electron multiplier - limited life, linearity issues, high-precision, signals < 2e6 cps
Popular combinations Gas source 1) Dual inlet isotope ratio mass spec (at GT, Lynch-Steiglitz and Cobb) - O, C, H ratio analyses 2) Elemental analyzer IRMS (at GT, Montoya) - N, C, S ratio analyses 3) Gas chromatograph IRMS (at GT, Chemistry) - compound-specific ratio analyses Solid source 1) Thermal Ionization mass spec (multi-collector) - heavy metals, REE ICP 1) ICP quadrupole mass spec (at GT, Taillefert) - trace metal analysis 2) Single collector magnetic sector ICPMS - higher-precision trace metal analysis 2) Multi-collector ICPMS (nearest at USC) - U/Th dating, TIMS replacement Micromass IsoProbe - MC-ICPMS
Inductively Coupled Plasma Mass Spectrometry detector Shared components of all ICPMS machines mass/charge discriminator high vacuum 10-7 bar sample cone skimmer cone “fore” vacuum 10-4 bar instrument housing atmospheric pressure
Quadrupole ICPMS • - measure concentrations • as low as several ppt • - no fuss sample preparation • (dissolve in 5% HNO3) • - get beam intensity • vs. mass/charge ratio or magnetic sector Faraday cup and ion counter (electron multiplier)
The torch box of an Agilent 7500 ICPMS spray chamber Ar feed torch RF coil The sample cone isolates the torch from the interior.
ICPMS plasma torch schematic plasma components
High-resolution ICPMS 2. High resolution ICPMS aka double-focusing ICPMS aka magnetic sector ICPMS - same front end as Q-ICPMS - combines magnet w electrostatic analyzer electrostatic analyzer separates ions by charge magnet separates ion by mass Faraday cup and EM
Multi-collector ICPMS 3. MC-ICPMS - same front end as other ICPMS - same magnet and ES as HR-ICPMS - multiple detectors spaced 1amu apart enable simultaneous measurement of many (~7) isotopes -good for what kinds of systems?
Low vs. High – resolution ICPMS and Interferences 56Fe very low concentrations in environmental samples, but high interest (why?) Unfortunately, 56Fe has the same atomic wt as ArO (40Ar+16O) Quadrupole measurement = INTERFERENCE! HR-ICPMS measurement = can distinguish 56Fe from ArO NOTE: most elements can be distinguished with a low resolution quadrupole
The importance of standards in mass spectrometry ICPMS: Can determine concentration to ~1% R.E. using calibration curve (below) Can monitor Sensitivity (signal response for given solution concentration) over time unknown sample = 8.2e7 cps, conc ~ 10.5ppb
REMEMBER: all mass spectrometers are “black boxes” we really have no idea what goes on from sample container to detector signal • Ex: you measure a count-rate of 10,000 cps for a given element, but you need to know how many atoms of that element, or its concentration, were in your sample • measuring isotope ratios is a powerful approach because we can measure • samples against standards with known isotopic ratios (it’s much more difficult to change a material’s isotopic ratios than it is to change its elemental concentration!) • isotope dilution takes advantage of ability to precisely measure ratios • ALL measurements need to include blanks and standards (either concentration or ratio standards)
Isotope dilution principle Isotope dilution is an analytical technique used in combination with mass spectrometry to determine the concentration of element x in unknown samples. ex: Rb A known amount of “spike” with known elemental concentration and isotopic abundances (what’s the diff?) is added to sample with unknown elemental concentration but known isotopic abundances. • Requirements: • The sample has natural (or known) isotopic abundance (usually true). • The spike and sample isotopic ratios are different.
More Commonly used ICPMS terms Nebulization efficiency – the amount of solution that reaches the plasma (~1%) - varies with sample matrix - surface tension, viscosity, and density of solution will affect neb. eff. - usually all standards, spikes, and samples are introduced as 2-5% HNO3 - an acid solution reduces complexation, surface adsorption Matrix effects – the changes in ICP characteristics with variable matrices - largely black box (we see these effects, cannot wholly explain/predict them) - you must carefully match the matrices of your standards/samples to obtain quantitative results Ionization efficiency – the amount of ions produced per atoms introduced - depends on matrix, focusing of beam through cones, lenses - usually no better than 1/1000
ICP detection limits for a variety of elements ICP-OES ICP-MS Detection limit – defined as 3 x the S.D. of the signal as the concentration of the analyte approaches 0 (measure stability at a variety of conc’s, extrapolate to 0; or measure 5% HNO3 blank solution)
Perkin Elmer Quadrupole ICPMS Instrument Detection limits, 3σ
Ion microprobe (or Secondary Ion Mass Spectrometry SIMS) • use an ion beam (usually Cs+1) to “sputter” a sample surface; secondary ions fed into mass spec 20μm
Accelerator Mass Spectrometry The AMS at University of Arizona (3MV) • prior to AMS samples were 14C-dated by counting the number of decays • - required large samples and long analysis times • -1977: Nelson et al. and Bennett et al. publish papers in Science demonstrating • the utility of attaching an accelerator to a conventional mass spectrometer • Principle: • You cannot quantitatively remove interferring • ions to look for one 14C atom among several • quadrillion C atoms. • Instead, you • destroy molecular ions (foil or gas) • filter by the energy of the ions (detector) • to separate the needle in the haystack. The AMS at LLNL (10MV)
c) ACCELERATOR generates 2.5 million volts, accelerates C- ions b) INJECTOR MAGNET separates ions by mass, masses 12, 13, and 14 injected d) TERMINAL C- ions interact with ‘stripper’ gas Ar, become C+ ions, molecular species CH destroyed e) ELECTROSTATIC DEFLECTOR specific charge of ions selected (3+) a) ION SOURCE generates negative carbon ions by Cs sputtering f) MAGNETIC SEPARATION 13C steered into cup, 14C passes through to solid detector g) Si BARRIER DETECTOR pulse produced is proportional to the energy of ion, can differentiate b/t 14C and other ions count rate for modern sample = 100cps http://www.physics.arizona.edu/ams/education/ams_principle.htm
Hurdles in mass spectrometry 1) Abundance sensitivity - ratio of signal at mass m to signal at m+1 - better with better vacuum - acceptable values: 1-3ppm at 1amu 2) Mass discrimination - heavier atoms not ionized as efficiently as light atoms - can contribute 1% errors to isotope values - can correct with known (natural) isotope ratios within run, or with known standards between runs
Hurdles in mass spectrometry (cont.) 3) Dark Noise - detector will register signal even without an ion beam - no vacuum is perfect and - no detector is perfect - must measure prior to run to get “instrument blank” if needed 4) Detector “gain” - what is the relationship between the electronic signal recorded by the detector and the number of ions that it has counted? - usually close to 1 after factory calibration - changes as detector “ages” - must quantify with standards Cardinal rule of mass spectrometry: Your measurements are only as good as your STANDARDS! Standards (both concentration and isotopic) can be purchased from NIST