AIR PRESSURE
AIR PRESSURE. Keeping an Atmosphere. Atmosphere is kept by the world’s gravity Low mass (small) worlds= low gravity =almost no atm. High mass (large) worlds = high gravity = thick atm.

AIR PRESSURE
E N D
Presentation Transcript
Keeping an Atmosphere • Atmosphere is kept by the world’s gravity • Low mass (small) worlds= low gravity =almost no atm. • High mass (large) worlds = high gravity = thick atm. • Gravity and pressure • Air pressure depends on how much gas there is i.e. The atmospheric thickness.
Gravity and Atmospheric Pressure • The stronger the gravity, the more gas is held by the world and the greater the weight of atm. on a point
Earth’s Atmosphere • About 10 km thick • Consists mostly of molecular nitrogen (N2) and oxygen (O2)
Gravity pulls the air molecules toward the earth, giving them weight. The weight of the air molecules all around us is called the air pressure.
High altitudes = lower pressure Low altitudes = higher pressure
Atmospheric Pressure Gas pressure depends on both density and temperature. Adding air molecules increases the pressure in a balloon. Heating the air also increases the pressure.
Air pressure is equal in all directions. Pressure = force per unit area
Barometric pressure goes down. As elevation goes up This is an inverse relationship.
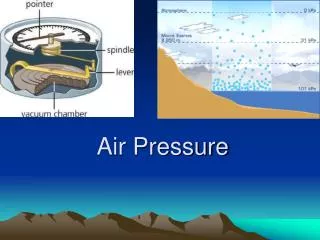
A Barometer is used to measure air pressure.
Torricelli’s barometer used a glass column suspended in a bowl of mercury. The pressure of the air molecules pushed the mercury up into the glass tube. The weight of the mercury in the tube was equal to the weight of the air pressing down on the mercury in the dish.
As atmospheric pressure increases… The mercury in the tube rises.
The Mercury Barometer Good: Bad: • Simple to construct • Highly accurate • Glass tube is fragile • Mercury is very toxic!
The AneroidBarometer • No fragile tubes! • No toxic chemicals! • No batteries! • Never needs winding!
MILLIBARS An aneroid barometer uses a cell which has had most of the air removed. As the air pressure around the cell increases, it presses on the cell, which causes the needle to move. Television weather forecasters usually give barometric pressure in inches of mercury. However, meteorologists measure atmospheric pressure in millibars.
Two types of barometric pressure measurements: Station pressure is the actual pressure at the recording location. It is affected by the local altitude. Station pressure on a mountain top will be lower than station pressure in a valley. Scientists need a fixed point of reference in order to compare barometer readings in different locations. That is why barometer readings are sometimes adjusted for elevation above sea level at the station location. Sea level pressure is referenced to sea level, so it has the same altitude anywhere in the world.
Most aneroid barometers have a needle which can be set to remember the previous reading.
Changing Pressure A rising barometer = increasing air pressure. This usually means: Rising barometer readings indicate that a high pressure system is approaching. Higher atmospheric pressure is usually associated with fair weather and clearing skies.
Changing Pressure A falling barometer = decreasing air pressure. This usually means: Falling barometer readings usually indicate the approach of an area of low pressure. Low pressure readings are usually associated with storm systems. Tornadoes and hurricanes can produce very low barometric readings.
6-1 • Fluids (air and water) flow from areas of high pressure to areas of low pressure. • Change in pressure across a horizontal distance is a pressure gradient. • Greater the difference in pressure and the shorter the distance between them, the steeper the pressure gradient and the stronger the wind. • Movement of air across a pressure gradient parallel to Earth’s surface is called a wind and winds are named for the direction from which they come.
Isobars in millibars, the closer the isobar the stronger the winds Rain Low Pressure High Pressure
The Atmosphere in Motion • Atmospheric pressure is a measure of the force pressing down on the Earth’s surface from the overlying air. • Pressure is often measured in different units including: • atmospheres (1 atmosphere is the average atmospheric pressure at sea level), • millibars (1 atmosphere = 1013.25 millibars), • pounds per square inch or psi (1 atmosphere = 14.7 pounds per square inch), • mm or inches of mercury (1 atmosphere = 760 mm or 29.92 inches of mercury) • torrs (1 torr = the pressure exerted by 1 cm of mercury). • Low air density results in rising air and low surface pressure. • High air density results in descending air and high surface pressure.
The Gas Law • Ideal Gas follows kinetic molecular theory, made up of large number of molecules that are in rapid random motion following perfect elastic collitions losing no momentum • How the Kinetic Molecular Theory Explains the Gas Laws • The pressure of a gas results from collisions between the gas particles and the walls of the container. • Each time a gas particle hits the wall, it exerts a force on the wall. • An increase in the number of gas particles in the container increases the frequency of collisions with the walls and therefore the pressure of the gas. • Avogadro's Hypothesis • As the number of gas particles increases, the frequency of collisions with the walls of the container must increase. • This, in turn, leads to an increase in the pressure of the gas. • Flexible containers, such as a balloon, will expand until the pressure of the gas inside the balloon once again balances the pressure of the gas outside. • Thus, the volume of the gas is proportional to the number of gas particles.
The Gas Laws • Charles Law • The volume of a gas increased with the temperature • The volume of a given amount of dry ideal gas is directly proportional to the Kelvin Temperature provided the amount of gas and the pressure remain fixed. • When we plot the Volume of a gas against the Kelvin temperature it forms a straight line. • V1 / T1 = V2 / T2 • Boyle’s Law • the product of the pressure and volume are observed to be nearly constant. • The product of pressure and volume is exactly a constant for an ideal gas. • p * V = constant