VSEPR Theory
VSEPR Theory. Bellwork 04/25. Draw the Lewis dot structure of H 2 S. What kind of bonding does this compound have?. VSEPR Theory. V alence S hell E lectron P air R epulsion Theory

VSEPR Theory
E N D
Presentation Transcript
Bellwork 04/25 • Draw the Lewis dot structure of H2S. What kind of bonding does this compound have?
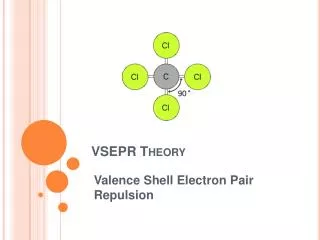
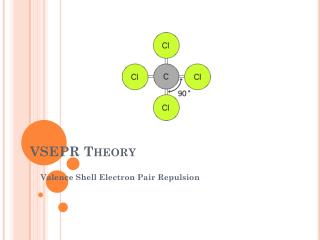
VSEPR Theory • Valence Shell Electron Pair Repulsion Theory • the repulsion between electron pairs causes molecular shapes to adjust so the valence electron pairs stay as far apart as possible • “Same charges repel!!” • Atoms exist at fixed angles to each other bond angle (e- always want to maximize this angle!) • Example: CH4
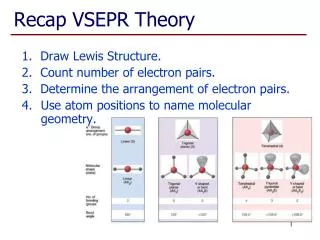
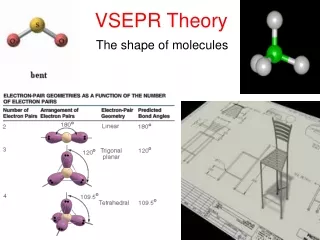
How to Predict Shapes • Draw Lewis dot structure of compound • Check valence electrons and bonds • Make sure to include all lone pairs of e- • Especially on central atom!! • Identify the shape of the molecule
Linear • Br2
Linear • CO2
Trigonal Planar • BF3
Bent • H2O • 3 atoms…. • Why not linear?
Pyramidal • NH3
Tetrahedral • CH4
Trigonal Bipyramidal • PCl5
Octahedral • SF6