Ch. 15 Benzene Reactivity
220 likes | 1.05k Views
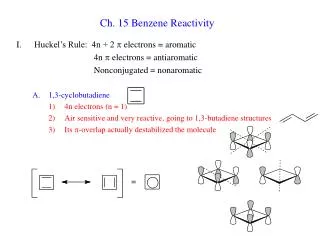
Ch. 15 Benzene Reactivity. Huckel’s Rule: 4n + 2 p electrons = aromatic 4n p electrons = antiaromatic Nonconjugated = nonaromatic 1,3-cyclobutadiene 4n electrons (n = 1) Air sensitive and very reactive, going to 1,3-butadiene structures

Ch. 15 Benzene Reactivity
E N D
Presentation Transcript
Ch. 15 Benzene Reactivity • Huckel’s Rule: 4n + 2 p electrons = aromatic 4n p electrons = antiaromatic Nonconjugated = nonaromatic • 1,3-cyclobutadiene • 4n electrons (n = 1) • Air sensitive and very reactive, going to 1,3-butadiene structures • Its p-overlap actually destabilized the molecule
The destabilization can be seen in the its rapid Diels-Alder reaction where it behaves as either the diene or dienophile • 1H NMR is very unlike benzene • 1,3,5,7-cyclooctatetraene • 4n electrons (n = 2) antiaromatic • Reactive like normal polyene • 1H NMR like alkene (5.68 ppm) • Structure is not planar or symmetric
Aromatic Cyclic Polyenes • [18]annulene has 18 p-electrons (n = 4 in 4n +2) • (CH)x gives the name of cyclic polyenes • Benzene = (CH)6 = [6]annulene • Cyclobutadiene = (CH)4 = [4]annulene • Planar molecule with equivalent C—C bonds • Stable with an benzene-like NMR • MO Explanation of Huckel’s Rule • Extended cyclic p systems have similar MO diagrams • Highest and lowest are not degenerate, all middle MO’s degenerate • 4n electrons does not fill the bonding MO’s (not stabilized) • 4n + 2 electrons does fill all bonding MO’s (stabilized)
Charged Molecules follow Huckel’s rule • 1,3-cyclopentadienyl anion is aromatic • Cyclopentadiene is not fully conjugated • The CH2 group has a pKa of 16 (very acidic for carbon) • The resulting anion is conjugated and aromatic (6 p electrons) • The cation is antiaromatic and very unstable • Cycloheptadienyl Cation is aromatic • Loss of a hydride in a reaction with bromine easily forms the cation • The cation has 6 p electrons and is aromatic
Cyclooctatriene Dianion is aromatic • [16]annulene cation and anion are aromatic
Electrophilic Aromatic Substitution • Reactivity of Benzene • Benzene is quite unreactive, but can be attacked by electrophiles • Electrophiles substitute themselves for one of the ring H’s • The double bonds (aromaticity) are not disturbed • Under the same conditions, a conjugated polyene would polymerize • Mechanism of Electrophilic Aromatic Substitution • E+ attacks benzene p-cloud forming a cationic intermediate • Intermediate loses H+ to regenerate the aromatic ring, now substituted
Step 1 is endothermic. The cation is less stable than the aromatic. • Addition of X- at this point would be a normal alkene addition, but would produce a nonaromatic product 5) Loss of H+ is favored (exothermic) because in reforms the aromatic ring
Halogenation of Benzene • Benzene is unreactive with X2 alone (not electrophilic enough) • A Lewis Acid catalyst activates X2 to become more electrophilic • Mechanism • For Br2, DH = -10.5 kcal/mol • F2 reaction is explosive • Cl2 reaction exothermic; use AlCl3 or FeCl3 as catalyst • I2 endothermic
Nitration • Benzene is unreactive towards HNO3 until H2SO4 activates it • Nitronium ion (NO2+) can react with benzene • Sulfonation • Benzene will not react directly with sulfuric acid • Fuming sulfuric acid contains 8% SO3
Mechanism • This reaction can be reversed. • The hydration of SO3 to H2SO4 is very exothermic, so SO3 reforms to undergo this reaction. • We can use sulfonation to block, then take it off to do another reaction • Benzenesulfonic acids have had many uses: • Detergents • Good Leaving groups for SN1 and SN2 reactions • Sulfonamide Antibiotics
Friedel-Crafts Alkylation • We need a reaction to form Aromatic C—C bonds • The Friedel-Crafts reaction gives us that power • Reactivity of alkyl halides: RI < RBr < RCl < RF • Lewis acids we can use: BF3, FeCl3, AlCl3, AlBr3 • Mechanism • Intramolecular Friedel-Crafts Reactions are possible
Any carbocation is susceptible to Friedel-Crafts reaction • Limits of the Friedel-Crafts Reaction a) Polyalkylation is difficult to stop (more than one R group adds)
Skeletal rearrangements of Alkyl groups frequently occur • We need to find a better way to make aromatic C—C bonds • Friedel-Crafts Acylation • Acyl Halides (and anhydrides) can add to benzene similarly to alkyl halides • Carboxylic Acids can be turned into acyl halides
Carboxylic acids plus acyl halides can form anhydrides • Both acyl halides and anhydrides give Acylium Ions in reaction with Lewis Acids • Acylium Ions can do electrophilic aromatic substitution • Advantages of Acylation: a) There is no danger of rearrangement Acyclium Ion
The electron withdrawing nature of the carbonyl group deactivates the benzene ring, so it won’t undergo multiple substitutions. • Lewis acid complexation increases the electron withdrawing ability of the carbonyl, further preventing a second reaction. • One full equivalent of AlCl3 is needed (not catalytic) • Must have aqueous workup to release product