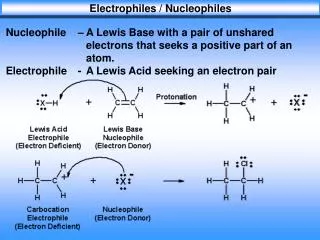
Nucleophile – A Lewis Base with a pair of unshared electrons that seeks a positive part of an atom. Electrophile - A Lew
Electrophiles / Nucleophiles. Nucleophile – A Lewis Base with a pair of unshared electrons that seeks a positive part of an atom. Electrophile - A Lewis Acid seeking an electron pair. Nucleophilic Substitution. Nucleophilic Substitution

Nucleophile – A Lewis Base with a pair of unshared electrons that seeks a positive part of an atom. Electrophile - A Lew
E N D
Presentation Transcript
Electrophiles / Nucleophiles Nucleophile – A Lewis Base with a pair of unshared electrons that seeks a positive part of an atom. Electrophile - A Lewis Acid seeking an electron pair
Nucleophilic Substitution • Nucleophilic Substitution • Reaction initiated by a Nucleophile (Lewis Base) which attacks an Electrophile, replacing a leaving group. - - • General Use • Substitution of Nucleophiles on primary and secondary haloalkanes. The Halide serves as the leaving group.
SN1 Reactions • SN1 – Substitution, Nucleophilic, Unimolecular • Substitution – Nucleophile substitutes for leaving group • Unimolecular • Rate of reaction is dependent on concentration of only one of the reactants • It is a first order reaction (sum of exponents in rate equation = 1). • Multistep reaction, where overall rate of reaction is determined by the slowest intermediate step.
SN2 Reactions • SN2 – Substitution, Nucleophilic, Multi-Molecular • Substitution in one step, where Nucleophile attacks carbon bearing the leaving group, which departs from back side of molecule. • A conversion of the molecular configuration occurs. • Multi-molecular - Reaction depends on the concentration of each of the reactants; thus the reaction is at least second order (sum of exponents in rate equation is >= 2).
SN1 Reactions • Carbocations • Intermediate Organic molecules formed in an SN1 reaction contain a trivalent carbon atom that carries a positive charge (electron deficient). • General structure is Trigonal Planar. • Relative stability related to number of Alkyl groups attached to positively charge Carbon atom.
SN1 Reactions • Alkyl groups are electron releasing The 2 electrons in filled sp3 orbitals of alkyl groups can overlap with empty p-orbitals of positively charged adjacent Carbon atoms that produces a stabilizing affect on the Carbocation (Hyperconjugation). • Electron density shifts toward the positive charge • The C-H & C-C orbitals adjacent to the unfilled p orbital of the Carbocation are filled. • Sharing of the electron density Delocalizes the positive charge, thus, stabilizing the system. • Tertiary Carbocations have 3 C-H or C-C bonds that can overlap the vacant p orbital producing more hyperconjugation than would 2 C-H or C-C bonds. • Thus, tertiary carbocations are more stable than secondary carbocations, which are more stable than primary carbocations.
SN1 Reactions • Stereochemistry • The Trigonal Planar structure of a Carbocation permits the Nucleophile to attack from either the front side or the back side. • A tertiary Carbocation substitution does not produce any stereo implications. • Some Carbocations, however, can produce different products from two reaction possibilities. • If one of the products is optically active and the other optically inactive, the reaction is said to have proceeded with Racemization.
SN1 Reactions • Racemization occurs when the reaction causes a Chiral molecule to be converted to an Achiral intermediate. • Chiral Molecule – Molecule that is not superposable on its mirror image. Chiral molecules possess handedness; thus, are capable of existing as enantiomers (stereoisomers of each other) • Achiral Molecule – Molecule that is superposable on its mirror image. Achiral molecules lack handedness, thus, are incapable of existing as enatiomers
Electrophilic Aromatic Substitution • Benzene, an aromatic structure, is generally unreactive (stable), but can be attacked by strong Electrophiles. • The stability of the aromatic ring ( bond structure) results in substitution as opposed to addition of the Electrophile as seen in the bond structure of Alkenes and Alkynes. • Two Step Process: • Electrophile (E+) attacks bond of ring and forms a cationic (positively charged) intermediate - Carbocation. • The Carbocation (an Arrhenium Ion) is not aromatic. • The positive charge is delocalized. • The formation of the C-E bond results in an sp3 hybridized carbon in the ring, interrupting the cyclic conjugation. • This arrangement is not thermodynamically stable. • A proton is lost from the ring regenerating the aromatic ring, which is stable.
Electrophilic Substitution Directing Effects on Aromatic Ring • When substituted aromatic rings undergo attack by an Electrophile (positively charged Lewis Acid), the group(s) already on the ring affect both the rate of reaction and the Regioselectivity (orientation) of the site(s) of attack. • Thus, the original group on the ring determines where the attacking group will substitute on ring, i.e., the ortho (1,6 positions), para (4 position) or meta(3, 5 position) sites. • Deactivation: • If the substituted group withdraws (accepts) electrons from the ring: • The electron density of the ring is decreased, deactivating the ring. • Thus, the Energy of Transition is increased and • The Free Energy of Activation is increased, resulting in • The “High Energy Transition State” leading to the formation of the delocalized “Arenium Ion” (positively charged Carbocation) to become “Less Stable.” • Therefore, the reaction rate is decreased relative to the rate on an unsubstituted aromatic ring. • The resulting Resonance” forms of the “Arenium Ion” favor the substitution of the second group at the “Meta” position, i.e., position “3” relative to the original group, ex. 3-nitrotrifluoromethylbenzene.
Electrophilic Substitution Directing Effects on Aromatic Ring • Activation: • If the substituted group Donates (releases) electrons to the ring: • The electron density of the ring is increased, activating the ring. • Thus, the Energy of Transition is decreased and • The Free Energy of Activation is decreased, resulting in • The “High Energy Transition State” leading to the formation of the delocalized “Arenium Ion” (positively charged Carbocation) to become “More Stable.” • Therefore, the reaction rate is increased relative to the rate on an unsubstitued aromatic ring. • The resulting Resonance” forms of the “Arenium Ion” favor the substitution of the second group at the “Ortho/Para” positions, i.e., positions “1, 4, 6” relative to the original group, ex. Orthobromotoluene.
Electrophilic Substitution Directing Effects on Aromatic Ring • Arrhenium Ion - The reaction rate-determining step in Electrophilic Substitutions of substituted aromatic rings is the step that results in the formation of a Carbocation (Arrhenium Ion or Sigma Complex) If Q is an Activator - an electron-releasing (donating) group - relative to hydrogen, the reaction occurs faster than substitution on an unsubstituted ring. Resonance forms favor o,p-directing If Q is a Deactivator- electron-withdrawing (accepting) group – the reaction occurs slower than substitution on an unsubstituted aromatic ring. Resonance forms favor m-directing.
Electrophilic Substitution Directing Effects on Aromatic Ring • Regioselectivity – Substitution Orientation The Ortho/Para vs. Meta directing effect of the first substituted group on the ring is accounted for by the interplay of two factors working simultaneously, either of which can be dominate. Induction Effect / Resonance Effect • Inductive Effect: • An intrinsic electron-attracting or –releasing effect that results from a nearby Dipole. • Polarization is induced from electrostatic interaction of the bonds. • Governed by relative Electronegativity of the involved atoms. • If a substituted group is more electronegative than the carbon in the ring, then the ring is at the positive end of the Dipole. • Inductive effect tapers off rapidly with distance. • The ring can be “Activated” or Deactivated” through the Inductive Effect.
Electrophilic Substitution Directing Effects on Aromatic Ring • Induction – Activation by Electron Donation (Release) • Groups that donate (release) electrons to the ring by Induction are Activating. • The Methyl group (CH3) and other Alkyl groups donate electrons (through Hyperconjugation effect) that delocalizes (stabilizes) the positive charge on the intermediate Arenium ion ring making it more receptive to the positive charge of the attacking Electrophile. • The resonance forms resulting from O,P attack benefit from this stabilizing effect of the donated electrons forming a stable intermediate Carbocation. • In Meta attack, the resonance forms do not benefit from the stabilized configuration. • Thus, Ortho / Para products predominate.
Electrophilic Substitution Directing Effects on Aromatic Ring
Electrophilic Substitution Directing Effects on Aromatic Ring • Induction – Deactivation by Electron Withdrawal (Acceptance) • When substiuted on an aromatic ring, electronegative groups or elements more electronegative than carbon form a Dipole with the positive end of the Dipole attached directly to the ring. • An attack by a positively charged Electrophile attempts to put an additional positive charge on the ring that destabilizes the Intermediate Arenium ion (carbocation) resulting from the Electrophilic attack. The Meta attack, however, is less affected by this destabilization, thus, Meta substitution is more favored. • Electronegative groups or elements more electronegative than carbon attached directly to the ring are electron withdrawing.
Electrophilic Substitution Directing Effects on Aromatic Ring • Induction – Deactivation by Electron Withdrawal (Acceptance) (Con’t) • Groups that form Dipoles
Electrophilic Substitution Directing Effects on Aromatic Ring
Electrophilic Substitution Directing Effects on Aromatic Ring • Resonance Effect • Effect by which a substituent exerts either an electron-releasing or –withdrawing effect through the bond system of the aromatic ring. • Occurs through the Bonds • Occurs over Longer Range • Particularly Strong In “Charged” Systems • Presence of substituent may increase or decrease resonance stabilization of the intermediate Arenium ion
Electrophilic Substitution Directing Effects on Aromatic Ring • Resonance (con’t) • Groups that donate (release) electrons by resonance are Activating and they are O,P-directing • Benzene rings bearing, Methyl, Alkyl, Methoxy, Amino (NH2) and Hydroxyl (OH) groups are strongly activated. • They bear one or more nonbonding electrons pairs, which can participate in Resonance. • The Resonance form adds electron density to the delocalized ring lending extra stability to the intermediate Arrhenium ion. • Note: These groups are electron withdrawing from an inductive viewpoint (more electronegative than Carbon), but the Resonance Effect dominates making them Otho/Para Directing.
Electrophilic Substitution Directing Effects on Aromatic Ring
Electrophilic Substitution Directing Effects on Aromatic Ring • Resonance (con’t) • Groups that withdraw electrons by Resonance are Deactivating. • Groups bearing a polarized double or triple bond, whose positive end is attached to the benzene ring. • The Meta resonance forms of the substituted Benzene Ring do not attempt to place additional positive charge next to the Carbon atom attached to the Electron withdrawing group. • The Meta forms are less destabilized and therefore favor Meta substitution. • Note: These groups are strongly deactivating from both the Inductive Effect and the Resonance Effect and are Meta directing.
Electrophilic Substitution Directing Effects on Aromatic Ring
Electrophilic Substitution Directing Effects on Aromatic Ring • Induction and Resonance Effects Oppose Each Other • The net effect on whether the first substituted group acts as an O,P directing “Activator” (Electron Releaser) or a Meta-directing “Deactivator’ (Electron Withdrawer) in the Electrophilic Substitution Process depends on the relative dominance of: • Inductive Effect ( bond framework - Electronegativity) • vs. • Resonance Effect ( orbitals overlap aromatic system).
Electrophilic Substitution Directing Effects on Aromatic Ring • Summary – Induction / Resonance: Induction • An intrinsic electron-attracting or –releasing effect that results from a nearby Dipole. • Groups that release (donate) electrons by inductionare activating and they are o,p-directing. • Groups that withdraw (accept) electrons by induction are deactivating and they are meta-directing Resonance • Occurs through the Bonds • Groups that release (donate) electrons by resonance are activating and they are o,p-directing • Groups that withdraw (accept) electrons by resonance are deactivating and they are meta-directing
Electrophilic Substitution Directing Effects on Aromatic Ring Effects of Deactivating (Electron Withdrawing) Groups on the Orientation of Substituted Electrophiles CF3 is strongly electron withdrawing (deactivating) leaving the ring electron deficient with a developing positive charge at the end of the dipole, i.e., on the ring carbon atom adjacent to the Deactivating Group. This deactivates (destabilizes) the ring toElectrophilic substitution As the positively charge Electrophile (E+) attacks the Ortho/Para resonance structures it attempts to add additional positive charge to the carbon adjacent to the positively charged withdrawing group, further destabilizing the ring; thus Ortho/ Para substitution are not favored. In Meta attack the carbon atom bonded to thedeactivating CF3 group does not share as muchof the positive charge of the ring with the Deactivating Group, i.e., the positive charge is spread more evenly about the ring. Thus, the ring is less deactivated than the Ortho/Para attack and therefore is more susceptible to substitution at the Meta position.
Electrophilic Substitution Directing Effects on Aromatic Ring Effects of Activating (Electron Donating) Groups on the Orientation of Substituted Electrophiles The Amino group is a strong activating group. Even though the amino group is moreElectronegative than the attached Carbon theelectron releasing resonance effect is much more dominant that the Deactivatingelectron Withdrawing effect. In O or P attack the nonbonding pair ofelectrons from the Nitrogen can form, through resonance, an extra bond to the Carbon atom completing the outer octet of Electrons. This stabilizes one of the Arenium ion (sigma complex) resonance structures. In Meta attack of a ring with a substituted activating group, none of the resonance structures are able to take advantage of the resonance contribution from the non-bonding pair of electrons. Therefore, the structure remains less stable and less subject to substitution at the Meta Position.
Electrophilic Substitution Directing Effects on Aromatic Ring Ortho & Para Directors Meta Directors Activators (Donate, Release Electrons) • Available pair of unbonded electrons to donate to ring. • Protons on ring are more shielded • Tendency for decreased Chemical Shift • Deactivators (Withdraw, Accept Electrons) • No unbonded electron pairs • Less Shielding of Protons on ring • Tendency for increased Chemical Shift Methyl & Alkyl groups are activating because of the stabilizing effect of sp2 hybridization (hyperconjugation) of an unbonded electron in methyl radical. Halogens have a net withdrawing effect through induction, but there is sufficient competition from resonance to make them o,p directors.