Stellar Spectroscopy
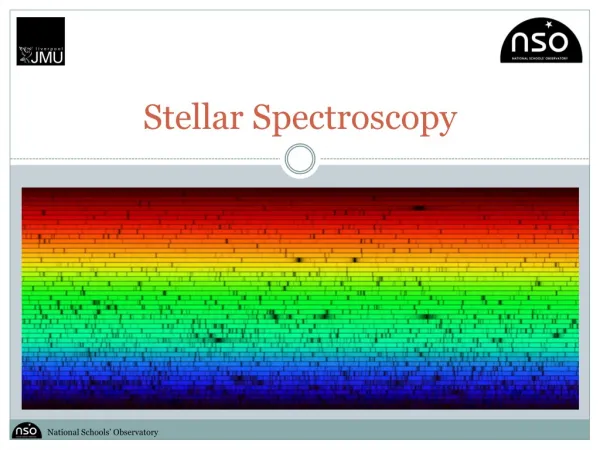
Stellar Spectroscopy. Introduction. Spectroscopy is a branch of science that is concerned with the investigation and measurement of the spectrum of light produced when matter interacts with electromagnetic radiation.

Stellar Spectroscopy
E N D
Presentation Transcript
Introduction • Spectroscopy is a branch of science that is concerned with the investigation and measurement of the spectrum of light produced when matter interacts with electromagnetic radiation. • In astronomy, spectroscopy is a very useful tool because telescopes can easily measure the spectra of far away objects, such as stars and galaxies. • The following activity will focus on stellar spectroscopy (the spectra of stars) and how those spectra can be used to classify stars.
Introduction • The intensity of those different wavelength components is then measured to create a spectra. The spectra can then be investigated in detail to discover useful information about the target. Rainbows are the spectra of our Sun. • The Liverpool Telescope (LT) has its own spectrograph – FRODOspec (Fibre fed Robotic Dual-beam Optical Spectrograph), and the following activity uses real data taken by the LT using FRODOspec. • We measure the spectra of luminous objects by attaching a special instrument to a telescope, known as a spectrograph. • Within the spectrograph, light is passed through a series of prisms and mirrors in order to split the light into its different component wavelengths (its spectrum).
FRODOspec Longer wavelength light is sent down the ‘red arm’. Shorter wavelength light is sent down the ‘blue arm’. The light from each arm is then split as it passes through a prism, and a detector captures the resulting spectrum on a CCD device. Light collected by the telescope enters the FRODOspec instrument and is split into two beams: The above image shows the inside of FRODOspec during its development.
FRODOspec Each of the vertical lines are repeats (or orders) of the same spectrum. You can see a line feature (circled red) running across those orders. In this activity you will be provided with a 2-D spectrum (wavelength versus intensity) that has been extracted from the raw data. The image to the right shows the raw spectrum of the star BD-02-5059.
Types of Spectra In general, there are two types of spectra that can be easily identified, EMMISION spectra and ABSORPTION spectra. An example of an emission spectra: An example of an absorption spectra:
Emission Spectra • Due to the temperature of the gas, the atoms within it will possess lots of energy, and some of their electrons will occupy excited states around the nucleus. • These electrons will de-excite (loose energy and fall to a lower energy level), with the lost energy being emitted as a photon at a specific wavelength. • Such processes cause bright lines in a spectra – Emission lines. Emission spectra occur when electromagnetic radiation originates from a cloud of relatively hot (compared to its surroundings) gas.
Absorption Spectra • Radiation at specific wavelengths from the star interacts with (is absorbed by) atoms in the cold gas, causing their electrons to gain energy and enter excited states. • These electrons quickly de-excite and emit photons at the same wavelengths. However, the direction of the emitted light is random and this leads to the appearance of dark lines (or missing light) in the resulting spectra, corresponding to the wavelengths that were absorbed by the gas. These lines are known as absorption lines. • Astronomers most frequently use absorption spectra to study stars. Absorption spectra occur when electromagnetic radiation from a background star passes through a relatively cold gas.
Absorption Spectra • Temperature • Rotational velocity • Translational velocity (overall motion of the star) • Density • Chemical composition • Metallicity (how many heavy elements stars contain) With careful analysis of the absorption lines, astronomers can learn many things about as star, including: Image of the Pleiades Star Cluster Some astronomers liken the spectrum of a star to a very basic DNA test that allows them to determine what kind of star the light originated from.
Chemical Composition Certain elements and molecules preferentially absorb and emit light at different wavelengths. It is possible to deduce from spectra which elements are causing lines at particular wavelengths, and hence determine the chemical composition of the object being observed. Analysis of a spectrum of the Sun led to the discovery of Helium, even before it was discovered on Earth! Above: Emission spectra of different elements.
Blackbody Radiation Before investigating real spectra, we need to talk about Blackbody radiation. • All stars are made up of a hot, dense interior and a cooler outer layer, or atmosphere. • The interior of the star causes a continuous spectra, known as a blackbody spectrum. • If there was no absorption or emission from the surrounding atmosphere, a stars spectrum would be close to a pure blackbody spectra. • The blackbody curve (see next slide) represents the spread of intensity of the radiation being emitted over all possible frequencies. • A metal glowing red hot in a furnace is an example of blackbody radiation. • The shape of the blackbody spectrum depends on the temperature of the body.
Blackbody Radiation Warmer objects have blackbody spectra that peak at shorter wavelengths, whereas cooler objects have blackbody spectra that peak at longer wavelengths.
Blackbody Radiation The dips in the curve (where it drops below the BB spectra) represent ABSORPTION lines. The peaks (where it rises above the BB spectra) represent EMISSION lines. In reality the measured spectra is never a perfectly smooth blackbody (BB) due to absorption and emission features. An example of a spectra showing absorption and emission features (Sloan Digital Sky Survey).
Wien’s Law • In order to classify a star, it is important for an astronomer to know the temperature of that star. • Stars range in temperature from about 2000K to 40,000K. • Using Wien’s law, it is possible to calculate the surface temperature, or ‘effective’ temperature, if we know the wavelength its blackbody curve peaks at. • where W is Wien’s constant and = 2.8977685×10−3 m·K. As an example, our Sun’s blackbody spectrum peaks at a wavelength of around 500 x 10-9 m, giving an effective temperature of about 5800K.
The Doppler Effect • One of these effects, the Doppler Effect, occurs due to an apparent shift in the wavelength and frequency of a wave due to relative motion between the source and the observer. • For example, we experience the Doppler Effect when an ambulance drives past with its sirens on: • As the ambulance (the source) approaches you (the observer), the sound wave is compressed (bunched up) and the resulting sound is higher pitched (shorter λ) • Once the ambulance has passed, the wave is stretched and the pitch sounds lower (longer λ) Shorter wavelength Longer wavelength
The Doppler Effect • The same effect is observed with light, when a star (the source) is moving towards or away from observers located on the Earth. • We can detect this motion in the spectrum of the star. • If the star is moving AWAY from the Earth, the light waves are stretched out and the whole spectra experiences a shift towards the red end of the spectrum – a red shift. • If the star is moving TOWARDS the Earth, the light waves are bunched up and the spectra experiences a shift towards the blue end of the spectrum – a blue shift. Shorter wavelength Longer wavelength
Spectral Lines • If there were no external factors at play, the absorption spectra of a star would show very thin spectral line features at the exact wavelengths corresponding to amount of energy transferred to the atom when one of its electrons has been excited to a higher energy level. • In reality, there are a number of different factors that act to spread out or blur the spectral lines across a wider range of wavelengths, a process called spectral line broadening.
Spectral Line Broadening • Temperature (Doppler Broadening) • Stellar Rotation • Pressure The images below are of the same absorption line feature, in this case due to hydrogen, as it appears in the spectra of three different stars. Spectral broadening can be caused by a number of different factors, including:
Doppler (Thermal)Broadening • Atoms in a gas move around at different speeds and in different directions . • This movement can cause light to be Doppler shifted as an atom in motion emits a photon, and because some of the atoms will be moving away from the observer and some moving towards, the overall effect will contain both blue-shift and red-shift components. • In a hot gas, the atoms possess more energy and thus move around more quickly than in a cooler gas. This means that Doppler broadening is more significant in hot stars. The Doppler broadening of spectral lines is the major factor is stellar spectra. Hot Star Cool Star
Rotational Velocity Stars are not just static objects as they appear in the night sky. They are made up of a fluid mass of gas and plasma (hot gas) that rotates around a common axis. The Sun rotates once every 24.5 days. When we obtain the spectra of distant stars, however, the light is gathered from across the entire disc of the star and not from just one point.
Rotational Velocity This means that some of the star is rotating towards us and some away. The parts rotating TOWARDS the observer are blue-shifted. The parts rotating AWAY from the observer are red-shifted. The net effect is that the star’s spectral lines appear to be smeared out, or rotationally broadened.
Spectral Classification • It is possible to use the absorption spectra of individual stars to classify them into distinct groups. • Astronomers classify them by temperature, into groups “OBAFGKM”, with group O containing the hottest and M containing the coolest. Note that there is a direct correlation between temperature and mass.
Spectral Classification • These groups are further divided into sub-groups numbered from 0 to 9, where 0 is the hottest; for example, B1 is hotter than B3, and F2 is hotter than F9. • The Sun has a surface temperature of 5800K, putting it in the G2 classification.
Activity Now you know a little more about stellar spectroscopy, you will soon get the chance to look at some real spectra from the Liverpool Telescope! Your teacher will supply you with an Excel file containing the 2-D spectral data for 9 stars, each under its own separate tab or page of the worksheet. You are required to plot the spectra for each of these stars. • Plot the number of counts against the wavelength for each star. You should produce spectra similar to the one seen below (don’t forget to label your axes appropriately). • The shape of each spectra will vary due to the spectral class of the star. Intensity The nine spectra cover many of the different classes of stars. Wavelength (Angstroms)
Activity • Once you have plotted the nine spectra, you can compare them to see how the different stellar classifications vary. Things to look out for are the width and depth of particular spectral lines. • The Hydrogen line at 6563 angstroms (10-10m) is a good absorption line for spotting any differences. • Hotter stars will have a wider absorption lines due to Doppler broadening. • Cooler stars tend to have more erratic spectra than hotter stars. • Some spectral lines will appear in some stars, but not in others. Intensity Make notes of your observations ! Wavelength (Angstroms)
Activity Once you’ve had a good look at the spectra of the 9 known star types, it’s time to use this knowledge to classify stars of an unknown star type. Your teacher will now supply you with another Excel file containing the spectra for two stars that you will have to plot and then classify. Using the information provided in this presentation, and by making comparisons to the spectra you have already plotted, try to classify the mystery stars. Good luck! p.s. If you are really stuck, your teacher will have the answer.