FDA
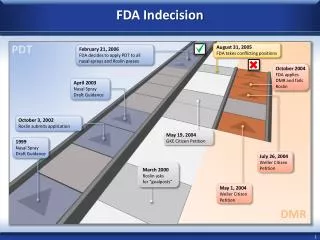
FDA. Import Process. Import Process. Entry Reviewer. IB. Request Documents. Pending FDA Review (FEX, SAM, DTR,DER). SAM. FEX. May Proceed. CB. IB Release. Detain. Release. Refuse. IMPORT PROCESS. Importer or agent files entry documents with Customs

FDA
E N D
Presentation Transcript
FDA Import Process
Import Process Entry Reviewer IB Request Documents Pending FDA Review (FEX, SAM, DTR,DER) SAM FEX May Proceed CB IB Release Detain Release Refuse
IMPORT PROCESS • Importer or agent files entry documents with Customs • FDA Reviews documents which include CBP 3461, invoice, bill of lading, packing list, etc. • FDA makes a determination on the admissibility of the product based on FDA regulation. • http://www.foodsafety.gov/~lrd/import.html (Good resource on FDA Import Procedures)
FDA ENTRY REVIEW • FDA Review Entry Documents to determine if a physical examination is needed. It’ll be helpful if the following information is provided (help speed up the entry review process) - LACF – Low Acid Can Food (SID/FCE information) (make sure to provide can dimension for every size can) - Drug Affirmation of Compliance - Device Affirmation of Compliance - Accession Number (RadHealth Product)
FDA FIELD EXAMINATION • FDA Sends an Notice of FDA Action to the Importer of Record, Consignee, and the Filer • The Importer of Record or Filer will need to submit a location letter to FDA that should include the following information: - Entry # - Name and address where product is held. - Dates when the entry is available for field examination. - Point of Contact (Name and Phone Number)
FDA FIELD EXAMINATION • The Notice of FDA Action indicates which items are “Pending FDA Review”. • Make sure that the products that are under “Pending FDA Review” status are held intact and have not been distributed until a written notice or an additional message has been received by FDA. • FDA Investigators will determine if a sample collection is needed base on: -Nature of the product - FDA priorities - Past history of the commodity
FDA SAMPLE COLLECTION • Once samples have been collected by FDA Investigators, they are sent to an FDA lab for analysis. • An Notice of FDA Action is generated and sent to the Importer and filer indicating which products have been sampled. • When the sampled products are still pending FDA Lab analysis, it is important that the product is still held intact and have not been distributed until FDA makes a final determination on the admissibility of the products. • If the sample was determined to be in compliance with FDA requirements, FDA sends a Release Notice to the importer and filer.
FDA DETENTION • If FDA detained a product, then an Notice of FDA Action is generated explaining the reason behind the detention. • Follow the instructions on the notice and pay attention to the timeframe for response. • You can contact the compliance status line for additional information regarding the detention.
Outline • Who Are Compliance Officers? • When Does The Compliance Branch Get Into “The Picture”? • What Do Compliance Officers Do? • What Do They Look For? • Actions Taken By Compliance • Import Alert Recommendations • Regulatory Actions • Other Compliance Actions • References
Who Are Compliance Officers? • They are Senior Consumer Safety Officers, Former Lab Analyst or HQ personnel. • They are Hearings Officers for FDA entries that are being Detained by the FDA District Office. • Have Authority to Detain, Refuse, Release or Investigate Entry Cases that have been assigned to them. • Compliance Officers (CO’s) refer cases to OCI, FDA Centers, FDA HQ, OGA’s and AUSA. • CO’s determine whether Regulatory actions can be taken on a case as it relates to the Food, Drugs and Cosmetic Act (FD&C Act) • It is their Responsibility to enforce the FD&C Act.
When Does The Compliance Branch Get Into “The Picture”? • After Field Exam Is Completed by Investigative Branches. • After The Analysis Is Completed by FDA Labs. • After Any Assignment Has Been Completed. • After Entry Review Has determined a detention is needed (Detention Request) • The Process For Compliance Starts when the Case from Investigations Branch has been forwarded to a Compliance Officer.
What Do Compliance Officers Do? (Process of Review) • Entry Documents * Flag discrepancies with entry information • Field Exam results • Analytical package from the lab when completed • Investigative Memo’s if discrepancies observed • Retail unit label & pictures of master cartons for any MISBRANDING Charges
What Does Compliance Look For? • Detention Without Physical Examination Criteria • DTR’s – Reason for Detention Request • FEX – Field exam class 3 findings * LBL Discrepancies * Invoice/Transmission/Actual Product Discrepancies * Filth Observations – held under insanitary conditions, filth, mold, seafood can defects, etc… • Analytical C3 Violative Findings • Memo for Redelivery, CBP Seizure, etc… • Label Review
Class 1 – Analytical Findings, FEX Usually - Released - Class 2 – Look In Historical Archive - Referred to the Centers For Concurrence - Detain Products with a reference to previous history. - Release Class 3 – ex: DEC, HIS, PES, MIC --- FEX - Take regulatory action - Set Up IB Work - Import Alert Recommendation Actions based on Classification
IA Recommendations • QA review of the file • Chain of custody and sample integrity. • Review analytical information • Prepare the cover memo and forward the recommendation to DIOP.
Label review [21CFR101] • Ingredient List • Common and usual name in English • Firm name and address • Net weight in U.S. measures • Nutritional Information
Prohibited ActsSec. 301 • The Act prohibits distribution in the US, or importation, of articles that are adulterated or misbranded. • The term “adulterated” includes products that are defective, unsafe, filthy, or produced under insanitary conditions • “Misbranded” includes statement, designs, or pictures in labeling that are false or misleading, and failure to provide required information in labeling.
Prohibited ActsSec. 301 The Act prohibits: • the distribution of any article required to be approved by FDA if such approval has not been given, 1. the refusal to provide required reports, and 2. the refusal to allow inspection of regulated facilities pursuant to Sec. 704.
Other Significant Compliance Actions • Reconditioning proposals • Follow-up to Refusals • Mitigations • Seafood HACCP Importer Verification • Consistent communication with Industry
Compliance References • FDA website: www.fda.gov • Import Compliance Programs. • Compliance Policy Guides. • Regulatory Procedures Manual; Chapter 9 • IOM • Import Alerts • Code of Federal Regulations • The Food, Drug and Cosmetic Act • www.cbp.gov